Work-related stress is widespread and a major source of sick leave (1). Research indicates that cognitive behavioral therapy (CBT) interventions are more effective than other treatments in reducing stress symptoms (2), but only few studies have included patients with clinical levels of stress symptoms. These studies have shown mixed results (2–5). In the current study, stress is understood in terms of Lazarus and Folkman (6), who define it as the subjective experience of demands that are not matched to the abilities of the individual, thereby posing a threat to his or her well-being. The term “work-related stress” refers to the experience of stress in relation to stressors at work, eg, high workload or interpersonal conflicts.
During the past decade, outpatient clinics of occupational medicine in Denmark have seen a rise in patients with work-related stress complaints. Many of these patients are on sick leave and diagnosed with adjustment disorder or reactions to stress [International Classification of Disease (ICD-10)] (1). Additionally, patients often complain of impaired sleep (5, 7) and cognitive difficulties like memory and concentration problems (5, 7, 8). Such complaints may interfere with work ability and influence sick leave duration (9). Although still uncommon in stress management intervention studies (SMT) (2) it therefore seems highly relevant to report on these outcomes.
Sleep, stress and work
Sleep problems are prevalent in modern society (10) and have been associated with a broad spectrum of negative health outcomes (11–19). Stress is a common self-reported cause (20). A review of early cross-sectional studies found a close association between psychosocial stress and impaired sleep, but research on the relation between real-life work stress and sleep has been sparse (21). A review by Kim & Dimsdale (22) found a small number of studies looking at the effect of daily life stress (occupational and non-occupational) on polysomnographic sleep measures, but results were inconsistent. A few prospective studies have found work-related stress in the form of perceived psychosocial work stressors to be predictive of development (23, 24, 25) and maintenance of sleep disturbance (25, 26). However, the relation between work stressors and sleep remains unclear. A recent review identified high job demands and low job control as predictors of reduced sleep quality, but effect sizes were small-to-moderate indicating that only a small proportion of the variance in sleep quality is influenced by work characteristics (15). Additionally, researchers have drawn attention to discrepancies between objective and subjective sleep measures and the risk of overestimating the effect of work stressors on sleep quality in studies using self-reported sleep measures (27, 28).
Work-related stress may be related to sleep problems through several mechanisms involving both physiological stress mechanisms and behavioral changes (10, 29). Psychologically the pivotal point is most likely related to thoughts and ruminations about work prior to sleeping, thereby contributing to continued arousal and wakefulness. Thus, Åkerstedt et al (20) found stress and worries at bedtime to be predictive of sleep quality on a day-to-day basis. In addition, the anticipation of disturbed sleep may also be a stressor in itself making it even more difficult to sleep.
Meta-analyses show that CBT targeted specifically at sleep problems is effective in treating a variety of sleep outcomes (30, 31). It is unclear whether interventions (like the current stress management intervention) that do not explicitly target sleep have a beneficial effect on sleep among stress patients with a clinical need (30). To the best of our knowledge, only one study has found improvements in self-reported sleep among stress patients on sick leave after a CBT-intervention containing some minor instructions on sleep. Apart from improving sleep habits, mechanisms of improved sleep by CBT could be related to reduced stress levels and improved coping strategies contributing to a reduction in rumination and physiological arousal before bedtime.
Stress and cognition
Forgetfulness and being unable to concentrate are common complaints among stress patients (5, 8, 32). Long-term exposure to stress has also been associated with cognitive impairments measured by neuropsychological testing (8, 32–38). Yet it is still unclear whether cognitive failures make one susceptible to stress or if stress causes the cognitive failures. Bridger et al (39, 40) found that self-reported cognitive failures were predictive of self-reported strain in the following two years. Furthermore, animal studies indicate that chronic stress affects several brain functions including memory (41, 42).
A few studies have examined cognitive impairments over time among patients with work-related stress complaints. One study found that self-reported cognitive failures improved after treatment (5) and former patients with burnout symptoms have shown improved cognitive performance after two years (43). These results indicate that cognitive performance improves when chronic stress levels are reduced either naturally or by treatment. Theoretically, mechanisms of improved cognition by CBT could involve improved coping behaviors and increasing the balance between stress exposure and rest. This could lead to decreasing levels of arousal contributing to cognitive problems.
Stress, sleep, and cognitive functioning
In several studies, psychological and physiological stress reactions have been the explanatory cause of both sleep problems and cognitive difficulties (5, 8). However, the relationships between stress and sleep, and between stress and cognition, may be bidirectional, since both impaired sleep and cognition could make the individual more susceptible to stress. Studies have also found a relation between experimentally induced sleep deprivation and impaired functioning across most cognitive domains (44–46). In the current study, sleep problems might therefore contribute to cognitive problems, but cognitive problems may also result in stress and rumination before bedtime, making it difficult to fall asleep. This interaction may be a contributing factor to the maintenance of work-related stress symptoms.
Study objectives
The current article is the first to report on the Danish randomized controlled trial (RCT) Behandling af Stress (BAS) [Treatment of Stress]. In the BAS trial, a cognitively based stress management intervention combined with a brief workplace intervention for patients on sick leave due to work-related stress was evaluated. The self-reported measures of sleep and cognitive failures in memory and distraction reported on in this paper were secondary outcome measures in the trial.
The primary aim of the current article is to examine a potential treatment effect on self-rated quality of sleep and self-rated cognitive failures in memory and distractions. A secondary aim is to evaluate the development of sleep problems and cognitive difficulties over time in a patient group with a clinical level of stress symptoms. It was hypothesized that the intervention group would experience a significantly larger improvement in sleep and cognition in comparison to the control group. It was also hypothesized that there would be some improvement over time in both groups and that there would be a significant positive association between symptoms of sleep problems and cognitive failures.
Study population and methods
Participants
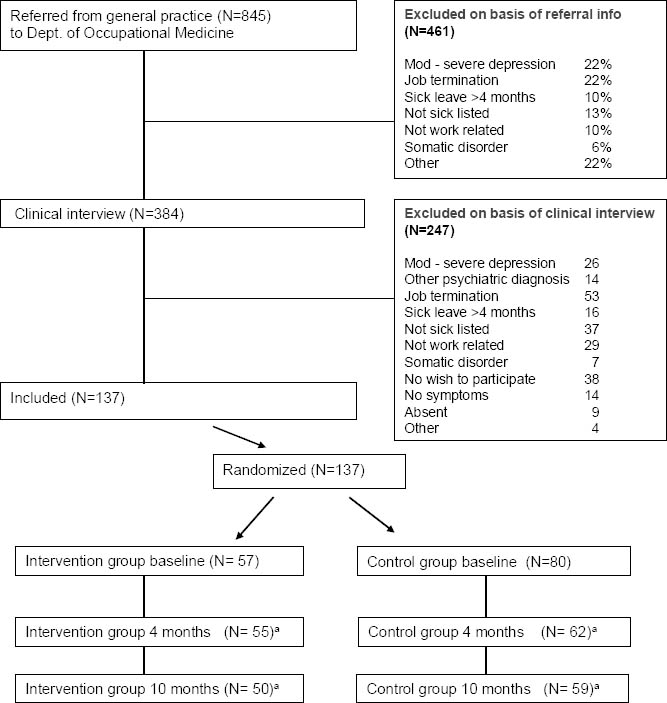
The RCT included 137 patients on sick leave (either full or part time) due to work-related stress complaints. Further information on recruitment, allocation, and outcome assessments is depicted in figure 1.
Eligibility/selection
Patients who, according to the statement of their general practitioner (GP), were referred to the Department of Occupational Medicine due to work-related stress were invited to participate in a clinical assessment interview with one of the psychologists from the BAS project to determine further eligibility.
Inclusion and exclusion criteria
Patients were included in the trial if they fulfilled the following criteria: (i) they had a diagnosis of adjustment disorder or reactions to stress [ICD-10 code: F43.2–F43.9 (not post-traumatic stress disorder)] or mild depression (F32.0) (47); (ii) they were on sick leave due to the above; (iii) the psychologist had evaluated their condition as primarily work-related; and (iv) they planned on returning to their workplace. Patients were cxcluded in the following cases: (i) comorbidity of another psychiatric illness (eg, moderate-to-severe depression); (ii) comorbidity of a recently emerged chronic somatic disease; (iii) pregnancy; (iv) substance abuse; (v) sick leave >4 months; (vi) any degree of disability pension; and (vii) fired or no wish to return to the work place.
The clinical assessment interview
Relevant GP-referred patients received a clinical interview to assess if the patient was suited for the study. The interview was based on an extended version of the normal procedure used in the clinic to evaluate and diagnose patients who are referred due to work-related stress complaints. Main areas covered were: employment history; present work situation, including work stressors and psychosocial work environment; the current situation related to sick leave; symptom development over time; and current symptoms. Non-work stressors, home life and dispositions for psychiatric illness were also covered. The interview aimed at an assessment of diagnosis and the likelihood of work-related stress having played a primary contributory role in symptom development. Uncovering the timing of stressor exposure and subsequent symptom development was an important aspect of the interview. Patients were not excluded if stressors related to non-work conditions were present, but these had to be of lesser significance.
Procedure
This article is part of the larger BAS study, which was designed as an RCT aimed at testing the efficacy of a CBT-based stress management intervention for patients on sick leave due to work-related stress. The current article reports on sleep and cognitive failures that were secondary outcomes in the BAS trial.
Randomization
If the person was accepted into the BAS study, the patient was randomized to either a treatment or control group. A secretary gave participants a randomization number from a list of 1000 randomly generated numbers between 0–100 000. Inclusion in the treatment group was based on the sum of the digits of this number (control group: unequal numbers, intervention group: equal numbers.). The random numbers were generated in a True Random Number Generator (www.random.org). Participants randomized to the control group did not receive further treatment from the clinic but were free to seek help outside of the trial.
Measurements
Baseline measurements and informed consent were carried out before the assessment interview and randomization procedure. Patients received information about the study and a comprehensive baseline questionnaire by mail. Thus, when answering the baseline questionnaire, patients were blinded as to whether they would be attending the treatment or control group. Follow-up questionnaires were answered at four and ten months after inclusion. Non-responders received two reminders
Intervention
The intervention comprised an individual CBT program as well as the offer of a brief workplace intervention. The individual cognitive intervention consisted of six one-hour sessions with a psychologist over the course of 16 weeks. Six sessions was chosen on the basis of previous research indicating that a limited number of CBT sessions in stress management interventions provided an optimal effect on psychological outcomes (2, 48). The intervention program was developed together with specialists in CBT and was designed specifically for patients on sick leave due to work-related stress complaints. The aims of the intervention were to: reduce the level of psychological and physical stress symptoms, strengthen the patient’s ability to cope with stressors at work, and identify and to help change work conditions if they were judged to be contributory factors. This involved the following aspects: (i) identifying work-related stressors or challenges; (ii) restructuring of cognitive and behavioral strategies contributing to the development of stress symptoms; (iii) psychoeducation about work-related stress; and (iv) homework assigned following each session.
Although the treatment was manualized, the psychologist had some flexibility in choosing techniques and homework assignments according to the needs of the patient. The manual contained a list and descriptions of specific CBT techniques that the psychologist was allowed to employ. Techniques not on the list could not be used. Decisions about which techniques to use were dependent upon clinical evaluations of the role played by dysfunctional assumptions, negative automatic thoughts, and so on. A process evaluation was conducted to ensure that treatment was conducted according to the manual. At the end of every session, the psychologist recorded on a checklist the techniques used in the session. Examinations of these data (not shown) indicate that certain techniques were nearly always employed. These included psychoeducation, problem and goal lists, the eliciting and restructuring of thoughts and assumptions, registration of symptoms. The treatment manual, in Danish, is available from the authors. A clinical psychologist with experience in CBT conducted all sessions.
The workplace intervention consisted of one or two meetings at the workplace with the patient, the psychologist, a leader, and/or other representatives from the workplace. The aim was to address stress-related problems at the workplace and facilitate a process that would meet the needs of the patient when he/she returned to work. Meetings typically lasted between 1–2 hours. The content of these meetings was dependent on the type of stressors present at the workplace, as well as the resources available to the employer. For example, being transferred, temporarily or permanently, to a different work group or department was sometimes an option at a large workplace, but often not an option at a small workplace. If stress-related problems concerned a high workload, unreasonable deadlines or ambiguous role definition, these were addressed by reducing workload or better specification of the work role. If stress was related to conflicts with colleagues, plans for dealing with these were discussed. The psychologist acted as an advisor to the workplace, but had no means of enforcing changes. The workplace meeting could take place at any time during the 16-week treatment period. Before such meetings were held, it was important for the psychologist and the patient to establish a common understanding of which changes were needed and realistically attainable. A key aim of these meetings was often to establish a better dialogue between the employer and employee with a focus on agreement about concrete steps that would be taken to ease return to work and reduce the likelihood of relapse. A gradual return to work, involving a period with part-time sick leave, was often the goal. When the psychologist could not participate (eg, because the patient preferred to discuss these matters in the workplace without the presence of a psychologist), the patient was aided in preparing for this dialogue.
Outcome measures
The Basic Nordic Sleep Questionnaire
Sleep quality was measured with the Danish version of the Basic Nordic Sleep Questionnaire (BNSQ) (49). The scale has been shown to have good internal consistency and has been used in a range of both epidemiological and clinical settings (49), but no reliability or validity data have been available so far. BNSQ consists of 27 different items in 21 questions. In the current study, 5 questions were chosen from the full scale: (i) Have you had difficulties falling asleep? (ii) How often have you awakened during the night? (iii) How often have you awakened too early in the morning without being able to fall asleep again? (iv) Do you feel excessively sleepy in the morning after awakening? and (v) Do you feel excessively sleepy during the daytime?
Participants were asked to answer according to their experience during the past four weeks. Questions were answered on a 5-point scale from 1 (never or almost never) to 5 (every night or almost every night). A Cronbach’s alpha of 0.68 was found for the selected scale.
The Cognitive Failures Questionnaire
The Cognitive Failures Questionnaire (CFQ) (50) was designed to measure everyday cognitive errors. Originally it was thought to measure the frequency of lapses in three areas: perception, memory, and motor function. Later studies have questioned this dimensionality (5, 51, 52). Wallace and colleagues (51–53) suggested a 4-factor solution with the dimensions memory, distractions, memory of names and blunders and found that this solution consistently provided a better fit than the 3-factor solution. The original questionnaire consists of 25 items, but in the current study we only used questions regarding memory and distractions since these dimensions were judged to be the most relevant for this group of patients. Respondents were asked to rate how often they experienced the described cognitive problems in the past four weeks and items were scored on a 5-point Likert scale ranging from 0 (never) to 4 (very often). In the present sample, a Cronbach’s alpha of 0.85 was found for the memory-scale and 0.85 for the distraction-scale.
Statistical analysis
Statistical analyses were conducted according to the intention-to-treat principle and thus included all data available. Statistical analyses were performed using the STATA (Stata Corp, LP, College Station, TX, USA) software package, version 11.2. Baseline characteristics in the two groups were compared using the chi-squared test of comparable distributions and Student’s t-test. Outcome analyses were performed with multivariate repeated measurements analysis. Due to lost-to-follow-up (see figure 1), the command ‘xtmixed’ in STATA was used as it handles missing data well. Model validation was performed using QQ-plots by group of residuals versus predicted values and residual probability plots. Single missing responses in a scale were replaced by the mean value of the remaining items on the relevant scale for each individual. Single mean responses were only calculated when >50% of all items in a scale were available. The relative use of single mean imputation was low.
Cohen’s d was used to measure the magnitude of treatment effects. Calculating Cohen’s d is a method often used to derive standardized mean differences on scales and questionnaires. Cohen’s d is measured by calculating the difference between values at baseline (T0) and follow-up (T4 and T10) divided by the combined standard deviation (SD) for the groups at baseline: d=mean(a) – mean(b)/(pooled variance of a and b). Results are interpreted according to following guidelines: small d< 0.2–0.5 SD; medium d=0.5–0.8 SD; and large d>0.8 SD (54). Effect sizes were calculated so that a negative difference was equivalent to an improvement or a reduction in symptoms. In addition to the intention-to-treat analysis, Spearman’s rho was conducted to test the relation between sleep and cognitive difficulties.
Results
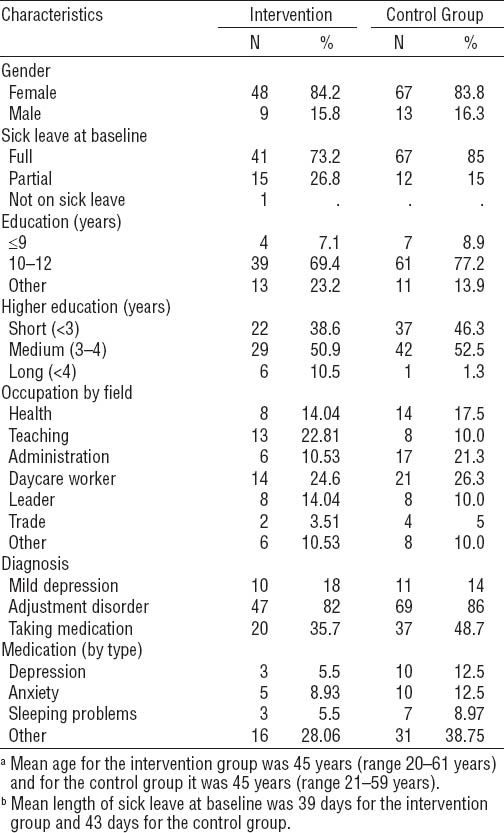
Patient characteristics of the intervention and control group are presented in table 1. No significant differences in baseline characteristics were found between the two groups. However, during the process of analyzing data, it was discovered that one person, who had previously been on sick leave had just resumed work when entering the intervention group. The analyses presented here include this person. Sub-analyses were conducted without the participant, but this did not alter the results for any outcomes.
Drop-out analyses were conducted of individuals who remained in and dropped out of the study. These groups were compared on demographic variables and baseline scores on sleep (BNSQ), cognitive failures in memory (CFQ-Memory) and cognitive failures in distractions (CFQ-Distractions). No significant differences were found on any of the variables between those who stayed and those who dropped out. Six treating psychologists participated in the study but sub-analyses showed no significant difference in treatment outcome.
Outcome measures between groups
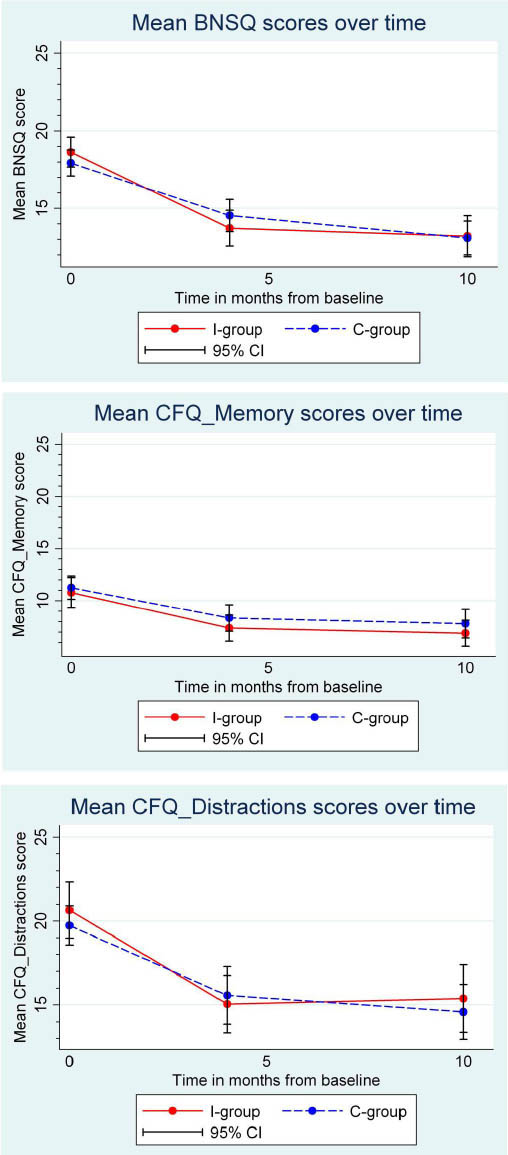
Changes in complaints are visually presented in figure 2. Outlined are changes from baseline to four and ten months follow-up on quality of sleep (BNSQ), cognitive failures in memory (CFQ-Memory), and cognitive failures in distractions (CFQ-Distractions). Visually, the curves of the intervention and control group seem close to identical for all three outcomes, but for BNSQ and CFQ-Distractions there seems to be a larger reduction in symptoms from baseline to four months follow-up than from the 4–10 months period.
Testing group differences on cognitive complaints and sleep problems
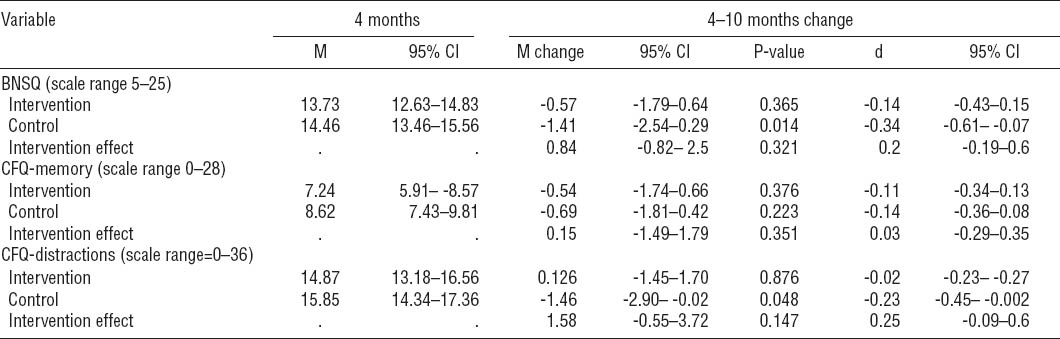
The results regarding group differences on all outcomes are shown in table 2, 3, and 4. Table 2 shows the baseline score, the mean change score at four months in absolute numbers followed by the corresponding P-value and effect size (Cohens’s d). Table 3 and 4 respectively show the mean change score, P-value and corresponding effect size for the change from 4–10 months and the overall change from baseline to ten months. At baseline, the two groups were comparable on all three outcomes. Looking at changes over time, no significant differences were seen between the two groups at the four or ten months follow-up. However, from baseline to four months follow-up there was a tendency for a larger improvement in the intervention group with regards to sleep problems and CFQ-Distractions. Although neither was significant, the intervention effect came close to significance depicting a small effect size on both sleep complaints and cognitive failures in distractions. This pattern was not repeated in the 4–10 months measurements. Results from the within group analyses are also shown in table 2, 3, and 4. Patients in both groups showed significant reductions on all outcomes during the first four months. Within group changes were medium to large on all outcomes, but a similar pattern did not appear in the 4–10 months period where improvements were generally small.
Table 2
Changes over time from baseline to 4-month follow-up. Effect sizes are depicted using Cohen’s d (standardized mean difference). [BNSQ=Basic Nordic Sleep Questionnaire; CFQ=Cognitive Failures Questionnaire; SD=standard deviation; 95% CI=95% confidence interval]
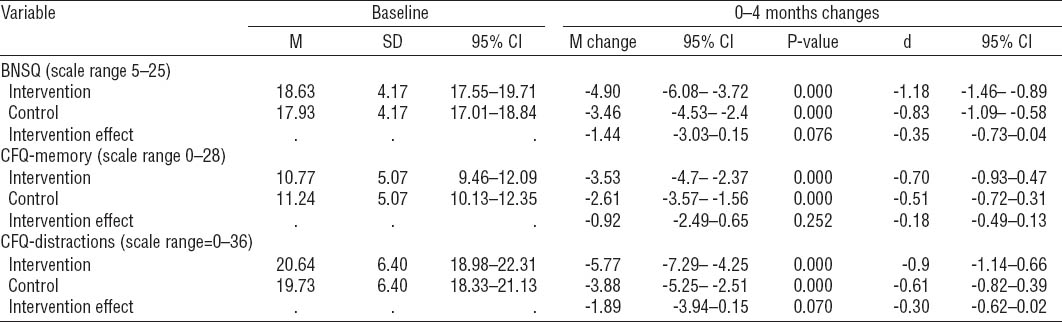
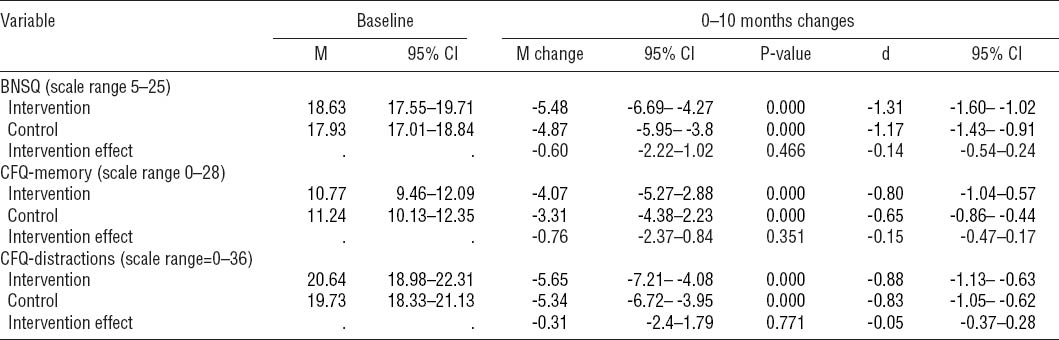
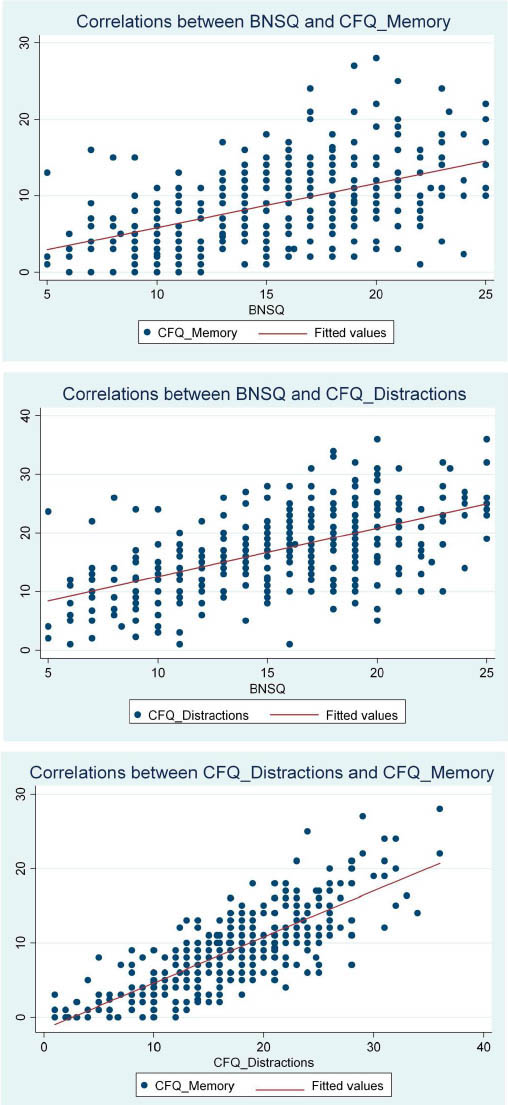
Intercorrelations between sleep and cognitive difficulties
As displayed in figure 3, there was a strong correlation between sleep difficulties and CFQ-Memory (r=0.5, P<0.0001) as well as between sleep difficulties and CFQ-Distractions (r=0.56, P<0.0001). CFQ-Memory and CFQ-Distractions were also highly correlated (r=0.8, P<0.0001).
Discussion
A stress management intervention comprising an individual CBT intervention and the offer of a small workplace intervention was not more effective than the control condition in reducing self-reported sleep problems and cognitive failures during ten months of follow-up. Tendencies toward a treatment effect were seen with regards to sleep and distraction at four months follow-up, but none were statistically significant at the chosen level of 95%. Cognitive failures in memory appeared less elevated at baseline and mean-changes may have been small after four months for this reason. The results were robust and remained unchanged also when conducted strictly by protocol without single mean imputation.
There may be several reasons for the lack of effect on sleep problems and cognitive failures. These outcomes represent somewhat specific stress symptoms that may vary greatly among stress patients, perhaps making it difficult to detect a treatment effect. As in other studies (4, 5, 55, 56), the chance of detecting a treatment effect in the current study may also be reduced by some degree of natural recovery in both groups. In addition, sleep problems and cognitive difficulties were not an explicit focus of the intervention. Therefore, directly addressing problems in sleep and cognition may be a way of improving future stress interventions. It could also be questioned if the treatment was extensive enough offering only six sessions of therapy. The choice of six sessions was based on reviews of stress management interventions, but the included studies in these reviews rarely (if at all) reported on sleep or cognition. It is possible that more sessions should be applied to target these outcomes.
In general, some remaining symptoms appear to be present at ten months follow-up with regards to both sleep and cognitive failures. We do not know if remaining symptom levels exceed the level of complaints in the general population. With regards to sleep, research has found sleep problems to be common within the general population especially among women and with increasing age (57). With regards to the CFQ, we were able to compare the follow-up scores on memory and distractions across groups at ten months follow-up (N=112) to the scores of a healthy control group of 60 healthy individuals used in another (as yet unpublished) stress study. The healthy group was highly comparable to our patient group with regards to age, education and gender distribution. When comparing the scores for memory and distractions at ten months follow-up in the current study with the healthy control group, we found that the patients have a higher score on both memory problems (mean difference=1.91 equivalent to a small effect size: Cohen’s d=0.42) and distractions (mean difference=4.58 equivalent to a moderate effect size: Cohen’s d=0.73). (Differences in group sizes were taken into account when calculating Cohen’s d.) Thus, it is possible that the remaining symptoms at 10 months follow-up still have implications in everyday life for the patients. Other studies have also found remaining symptom levels (within a variety of symptoms) above normal at the end of follow-up (4, 56, 58). It is currently unclear whether the decline in symptoms will continue after the first phase of recovery. It should be noted that we don’t know if elevated levels of cognitive failures were present before patients developed stress symptoms.
Apart from one comparable study by Willert et al (5), we did not find any other RCT that has investigated the effect of a stress management intervention on sleep and cognition (self-reported or objective measures). Some studies have investigated the development of sleep among patients with burnout using a matched control group (59, 60). Others have looked at the development of cognitive symptoms in burnout patients either naturally or after treatment (43, 60), but none of these studies involved a RCT design. These studies all show some degree of improvement over time in sleep physiology and self-reported cognitive failures, but no conclusions with regards to treatment efficacy can be made. It should be mentioned that most of these studies do not exclude patients with comorbid depression as we have done in the current study.
The study by Willert et al (5) found a significant treatment effect of a 3-month, group-based stress management intervention on self-reported sleep quality and cognitive failures. Effect sizes (Cohen’s d) were moderate ranging from 0.64 for sleep (seven questions from the BNSQ) and 0.57 for cognitive failures (the CFQ but measured as one factor and therefore hindering direct comparison with our study). The two studies differed in several ways. Contrary to the individual treatment program of the current study, Willert et al (5) evaluated a wait-list control design. Patients were on sick leave due to work-related stress complaints but had to have a plan of returning to work within four weeks. Willert et al (5) shortly addressed the area of sleep in the third session, where specific recommendations for insomnia and advice on healthy sleeping habits were introduced. The positive results of this study could indicate a beneficial effect of even a few sleep instructions in stress management interventions. The group setting could also have contributed to the positive results. In comparison to our study, the wait-list control design may have resulted in larger treatment effects since being on hold could delay expectations of recovery. Being in a wait-list control group may also prevent participants from seeking help on their own, which was often the case in our study.
In the current study, problems in self-reported sleep and cognition were primarily seen as stress symptoms, but some researchers have stated that changes in sleep physiology may not only be viewed as a symptom of stress exposure but also as part of the mechanism contributing to symptoms (59). The same could be said about cognitive problems. As suggested by Oosterholt et al (60), executive functions are essential in coping effectively with stressful work and may therefore play a role in the development of work-related stress disorders. Furthermore, although nothing can be concluded with regards to direction of causality, the highly significant correlation between sleep and cognition may support the assumption of an interdependent relationship between sleep and cognition. Thus, sleep impairment may independently contribute to problems in cognition which again may contribute to problems falling asleep and thereby create a negative spiral.
Strengths and limitations
The current study has several strengths. It is to the best of our knowledge the first RCT testing the efficacy of individual CBT in combination with a brief workplace intervention on sleep and cognition. Furthermore, the thorough selection process ensured that only patients with stress symptoms that were actually work-related were included. Unlike several other studies (2), patients were assessed to document a clinical need while patients with comorbid psychological illness were excluded.
However, the study also has several limitations. The design of a combined intervention of CBT and the possibility of a workplace intervention has been called for in systematic reviews (2, 48). But it was not possible to achieve a direct workplace intervention among more than 25% of the patients in the treatment group (maybe partly due to patients feeling stigmatized by bringing a psychologist into the workplace). In the remaining cases, the work focus of the intervention was limited to counselling of the patient.
Even though the participants were selected on the basis of a thorough screening interview, the group was still heterogeneous displaying a great deal of symptom variation ranging from mild symptoms of arousal, sadness, and problems in sleep and cognition to severe exhaustion, debilitating sleep difficulties, and cognitive problems. It should also be noted that the sample size was not based on power calculations on BNSQ or CFQ.
Another limitation involves the control group, which was free to receive treatment elsewhere (eg, psychological counselling). Sub-analyses showed that 41% of patients in the control group received ≥1 sessions with a psychologist outside the trial. Some patients also received offers within the Danish sick leave procedure. Only six patients in the control group did not report having received help by a psychologist, coach, psychiatrist, their doctor, or other sources of help. Naturally, this limits the chances of detecting a treatment effect and it would perhaps have been more reasonable to characterize the control group as a treatment-as-usual group. On the other hand, the possibility of seeking help elsewhere may strengthen the external validity of the study. Furthermore, the control group may have benefitted from the clinical interview at baseline, since the interview could have helped in clarifying issues related to stress symptoms and work conditions. Sub-analyses conducted after dropping those in the control group who had received sessions with a psychologist showed that the treatment effect at four months follow-up (that was previously borderline significant for sleep and cognitive failures in distraction) became significant for sleep (P=0.042 and Cohen’s d=- 0.4, 95%CI -0.78– -0.15) and for distractions (P= 0.035 Cohens d=-0.35, 95% CI -0.6– -0.025). At ten months, the groups no longer differed and there was no significant difference with regards to memory (P=0.45). The sub-analyses may indicate that the main results in this paper should be interpreted with caution especially with regards to underestimating the treatment effect.
There was a large amount of dropout in the control group (approximately 20%), but no differences were found between dropouts and non-dropouts on either demographic or baseline measures. It was by chance that the control group became larger than the intervention group. The true random numbers used in the randomizations process were not limited beforehand, thereby allowing one group to grow more rapidly.
In the current study we used only self-reported questionnaires. Subjective measures are sensitive to the psychological state of the patient and could influence outcome results. With regards to our measurement of sleep, the selected items from the BNSQ may be related to two different aspects of sleep. Items i–iii refer to complaints typically related to insomnia whereas the last 2 items (“sleepiness right after awakening” and “daytime sleepiness”) are related to sleepiness during daytime. Sleepiness may be understood as an increased tendency to doze off or fall asleep (61), but according to several researchers, insomnia may not be closely related to daytime sleepiness but rather increased fatigue (62, 63). The relation between sleepiness and fatigue also remains unclear (61). To ensure that our results were not affected by combining items on insomnia and sleepiness, we conducted sub-analyses removing the two items from the scale but this did not alter the results.
The items from the BNSQ did not contain information on napping and sleep length, which would have provided more detailed information about sleep quality. In addition future studies could benefit from objective sleep measures.
With regards to cognition, some studies have found that self-reported measures on cognition did not correlate with neuropsychological testing (60, 64). Therefore CFQ may not be a valid predictor of neuropsychological test scores but should perhaps be perceived as an indicator of how the individuals experience their cognitive capacity. This subjective experience may be highly influential on work ability and return to work.
Concluding remarks
A combined stress management intervention consisting of a 6-session CBT program combined with a brief workplace intervention did not reduce self-reported sleep problems or cognitive difficulties in the treatment group compared to the control condition. There was a tendency toward a treatment effect on sleep problems and cognitive difficulties, but none were significant. Furthermore, a substantial amount of recovery was found for both groups. The latter may be an important message for patients who have concerns about the recovery process regarding sleep and cognitive difficulties.