In 2007, the International Agency for Research on Cancer (IARC) concluded that shift work involving circadian disruption was probably carcinogenic in humans based upon evidence of an increased breast cancer risk among women. The IARC expert panel judged that there was “sufficient evidence” in experimental animals but “limited evidence” in humans (Group 2A) (1).
Possible mechanisms linking shift work and cancer center around the “light-at-night” (LAN) hypothesis, which postulates that exposure to artificial LAN suppresses pineal melatonin secretion and subsequently leads to an increase of sex hormones, which in turn could increase the susceptibility to hormone-dependent cancers (2). Also, anti-proliferative effects of melatonin that could influence tumor origin and progression have been demonstrated in vivo and in vitro for prostate cancer cells (3).
Although much of the research regarding the carcinogenicity of circadian disruption focused on breast cancer (4, 5), an increasing body of evidence has emerged associating prostate cancer with shift work. A meta-analysis of earlier research judged that the overall evidence was suggestive for a positive association between proxies of circadian disruption and prostate cancer risk (6). However, results from subsequent reports remained inconsistent (7–14).
Chronotype or diurnal preference may influence an individual’s sensitivity to circadian disruption (with “evening types” showing better adaptation to shift work) which in turn may affect cancer risks (15). However, previous study results on prostate cancer were not consistent with this hypothesis showing higher risks rather in evening types (13) or similarly increased risks for men with morning and evening, but not intermediate type (11). In addition, methods to assess diurnal preference differed between studies, which limits the direct comparability of results.
Therefore, we investigated the association of shift and night work with the incidence of prostate cancer using data of the population-based Heinz Nixdorf Recall Cohort Study (HNR) from the Ruhr area in Germany. In contrast to industry-based cohorts, we studied a variety of jobs associated with shift and night work, following recommendations by an IARC Working Group how to define shift work in epidemiological studies (16). We considered various non-occupational confounders that may affect the incidence of prostate cancer. In addition, we studied the differential effect of diurnal preference, assessed by preferred midpoint of sleep, on prostate cancer risk.
Methods
Study population
The detailed methods of the HNR study have been described elsewhere (17). Briefly, the HNR is a population-based cohort study involving a random sample of 45–74 year-old inhabitants of the highly industrialized Ruhr area in Germany. Participants for the baseline survey were recruited from 2000–2003 and a follow-up survey was conducted from 2011–2014.
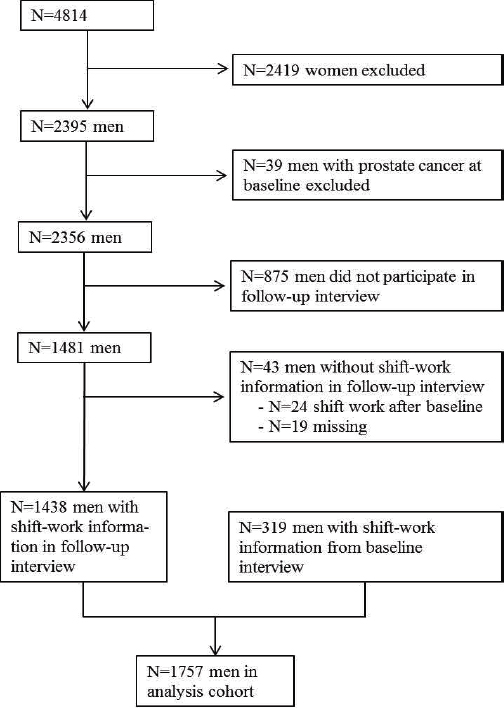
Of the 4100 men eligible to participate in the study, 2395 participated in the baseline survey. We excluded 39 participants who were diagnosed with prostate cancer before baseline. Of 2356 men without a history of prostate cancer at baseline, 1481 participated in the follow-up investigation. For 1438 men shift-work information was available from the follow-up interview. For an additional 319 men with insufficient shift-work information from the follow-up interview, shift-work exposure was complemented from the baseline interview. Therefore, 1757 men with information on shift and night work could be analyzed (figure 1).
All incident prostate tumors were monitored from baseline through September 2014. An independent endpoint committee of the HNR study evaluated patient records and, in the case of deceased participants, death certificates to assess the validity of the incident endpoints. Agreement of questionnaire-based incident cancer cases with medical records was evaluated for an 8-year follow-up and was considered to be satisfactory (18). The Ethics Committee of the University of Duisburg-Essen approved the study, and all subjects provided written informed consent.
Prostate cancer risk was higher among men who were not included in the analysis cohort (analysis cohort: 414.8 / 105 person-years versus 855.1 / 105 person-years). In addition, non-participants tended to be older (mean age 65.5 versus 57.5 years), had lower educational levels (66.1% versus 48% with education ≤13 years), and lower income (52.4 versus 33% in the lowest category).
Exposure assessment
For the follow-up interview, we employed a supplemental shift-work module, which included a detailed phase-by-phase assessment of the shift-work history and specific exposure characteristics, following recommendations by an IARC working group (16). Shift work was defined as any regular employment in shift systems including work hours outside 07:00–18:00 hours, whereas night work was defined as a shift that included work between 24:00–05:00 hours.
For subjects not participating in the follow-up, we used two questions from the baseline survey soliciting employment in shift work from all subjects who had been gainfully employed or unemployed for less than two years: (1) “Have you ever been employed in shift work?” (never; yes, not involving night work; yes, involving night work), and (2) “How long have you been involved in shift work?”
We included the following shift work variables in the analysis: 1) ever shift work (≥1 year) up to the baseline interview, 2) ever night work (≥1 year) up to baseline, 3) duration of shift work, and 4) duration of night work up to baseline, respectively. We quantified total duration of shift/night as follows: 0–<1, 1–<10, 10–<20 years, ≥20 years. To calculate duration of shift-work exposure, shift-work information from the follow-up interview was censored at baseline, ie, job periods involving shift work after baseline were not considered in the analysis. Information from the baseline interview up to the time of baseline was used for 319 men (figure 1).
Subjects were analyzed as having ever been employed in shift or night work during their work life. We also analyzed the association of employment in shift work without night work ever, although we observed this pattern in only 22% of shift workers,
To assess risk estimates for particular jobs associated with shift or night work, we studied subjects’ full job history. We analyzed the subject’s longest-held job (coded according to the “International Standard Classification of Occupations, revision 1968 (ISCO-68) that involved shift or night work according to the shift-work module. Due to the great variety of jobs in this population-based cohort (and the small number of single job titles), we present hazard ratios (HR) only for the three most frequent longest-held jobs in this cohort, as well as an analysis pooling jobs according to industrial sector: industries (eg, jobs in the metal, chemical, construction, electrical, and mining industry etc.) and public services (such as police officers, administrative and public transport employees).
To estimate a subject’s diurnal preference, we employed two questions from the follow-up interview soliciting the preferred bed- and wake-up time during days off work. From these two time specifications, we calculated the preferred midpoint of sleep. To define early, intermediate, and late midpoint of sleep, we determined age-specific tertiles separately in age groups 55–<60, 60–<65, 65–<70, 70–<75, 75–<80, and ≥80 years. Cut-offs at the 25% and 75% percentile were used to define early, intermediate, and late preferred sleep midpoint, resulting in age-specific sleep midpoints <02:30–03:00 hours for the early, 02:30–04:00 hours for the intermediate, and >03:30–04:00 hours for the late preference, respectively.
Total serum 25(OH)D at baseline was measured in 2007 on thawed samples using Liaison assay (DiaSorin). Vitamin D status was categorized as “low” or “high” based on a cut-off at the median concentration (15.3 ng/ml).
Statistical analysis
We performed Cox proportional hazards regression analysis with age at event as timescale to determine the association between shift work before baseline and prostate cancer incidence. We considered the age-at-event model as more suitable than the time-to-event model with linear age adjustment, because prostate cancer is a tumor of the elderly, and one would rather expect a difference between older and younger men than two subjects of the same age with different follow-up times. For the purpose of sensitivity analysis, we also calculated the Cox model with time to event as timescale and adjusting for age at baseline.
As potential confounders, we considered smoking status (never, former, current smoker), body mass index (<20, 20– <25, 25– <30, ≥30 kg/m2), physical activity [metabolic equivalent task (MET)-hours, assessed by an interview question regarding sports/physical activities during the last four weeks, combined with additional questions directed at duration and frequency of these activities, linear], alcohol consumption (g alcohol/week, linear), family history of prostate cancer, school education (≤13, 14–17 (reference), ≥18 years) adapted to the International Standard Classification of Education (ISCED), version 1997 (19), and equivalent income according to tertiles of income among men without prostate cancer [in German marks (DM)] [<2567.60; 2567.60–<3666.70 (reference); 3 666.70+ DM]. Next to family history of prostate cancer, potential confounders were included if risk estimates changed by >10% or if the P-value of the factor was <0.15.
We present three models: model 1, with age at event as timescale, an adjusted model including smoking status and family history of prostate cancer, and the fully adjusted model adding indicators of socioeconomic status (SES). Analyses were stratified by preferred midpoint of sleep and vitamin D-status. Tests for trend were calculated continuously.
Furthermore, we ran a sensitivity analysis restricted to men who had answered the supplemental shift-work module in the follow-up survey.
Results
We observed 76 incident primary prostate cancer cases during follow-up. The mean age at prostate cancer diagnosis was 66.8 years [standard deviation (SD) 6.83]. Among men without prostate cancer mean age was 77.3 years (SD 7.18) at the time of interview. At follow-up, 106 men without prostate cancer were ≥80 years (age at interview), whereas only one subject was ≥80 years at age of prostate cancer diagnosis. 34% of men reported a history of shift work, whereas a history of night work was indicated by 27%. The preferred midpoint of sleep was late for 14.1% and early for 13% of subjects. For further descriptive results see table 1.
Table 1
Descriptive data of the analysis cohort, stratified by shift work status at baseline, 1757 men. [DM=Deutchmarks)
| Total population | Prostate cancer | Never shift work | Ever shift work | |||||
|---|---|---|---|---|---|---|---|---|
|
|
|
|
|
|||||
| N | % | N | % | N | % | N | % | |
| Total | 1757 | 100 | 76 | 100 | 1156 | 100 | 601 | 100 |
| Shift work ever | ||||||||
| Never/less <1 year | 1156 | 65.8 | 38 | 50.0 | 1156 | 100 | ||
| Yes | 601 | 34.2 | 38 | 50.0 | 601 | 100 | ||
| Duration of shift work (years) a | ||||||||
| 0 –<1 | 1156 | 65.8 | 38 | 50.0 | 1156 | 100 | ||
| 1–<10 | 246 | 14.0 | 13 | 17.1 | 246 | 40.9 | ||
| 10–<20 | 127 | 7.2 | 8 | 10.5 | 127 | 21.1 | ||
| ≥20 | 219 | 12.5 | 17 | 22.4 | 219 | 36.4 | ||
| Missing | 9 | 0.5 | 9 | 1.5 | ||||
| Night work ever | ||||||||
| 0–<1 year | 1285 | 73.1 | 44 | 57.9 | 1156 | 100 | 129 | 21.5 |
| Yes | 472 | 26.9 | 32 | 42.1 | 472 | 78.5 | ||
| Duration of night work (years) a | ||||||||
| 0– <1 | 1285 | 73.5 | 44 | 57.9 | 1156 | 100 | 129 | 21.5 |
| 1– <10 | 204 | 11.7 | 11 | 14.5 | 204 | 33.9 | ||
| 10– <20 | 99 | 5.7 | 5 | 6.6 | 99 | 16.5 | ||
| ≥20 | 161 | 9.2 | 16 | 21.1 | 161 | 26.8 | ||
| Missing | 8 | 0.5 | 8 | 1.3 | ||||
| Age (years) | ||||||||
| 45–<55 | 700 | 39.8 | 16 | 21.1 | 453 | 39.2 | 247 | 41.1 |
| 55–<65 | 734 | 41.8 | 31 | 40.8 | 490 | 42.4 | 244 | 40.6 |
| 65–75 | 323 | 18.4 | 29 | 38.2 | 213 | 18.4 | 110 | 18.3 |
| Preferred midpoint of sleep a | ||||||||
| Early | 228 | 13.0 | 13 | 17.1 | 147 | 12.7 | 81 | 13.5 |
| Intermediate | 909 | 51.7 | 42 | 55.3 | 619 | 53.6 | 290 | 48.3 |
| Late | 248 | 14.1 | 8 | 10.5 | 166 | 14.4 | 82 | 13.6 |
| Missing | 372 | 21.2 | 13 | 17.1 | 224 | 19.4 | 148 | 24.6 |
| Family history of prostate cancer | ||||||||
| Yes | 23 | 1.3 | 2 | 2.6 | 13 | 1.1 | 10 | 1.7 |
| No | 1734 | 98.7 | 74 | 97.4 | 1 143 | 98.9 | 591 | 98.3 |
| Smoking status | ||||||||
| Never | 507 | 28.9 | 33 | 43.4 | 374 | 32.4 | 133 | 22.1 |
| Former | 809 | 46.0 | 34 | 44.7 | 506 | 43.8 | 303 | 50.4 |
| Current | 441 | 25.1 | 9 | 11.8 | 276 | 23.9 | 165 | 27.5 |
| Vitamin D status | ||||||||
| Low | 731 | 41.6 | 26 | 34.2 | 491 | 42.5 | 240 | 39.9 |
| High | 747 | 42.5 | 33 | 43.4 | 480 | 41.5 | 267 | 44.4 |
| Missing | 279 | 15.9 | 17 | 22.4 | 185 | 16.0 | 94 | 15.6 |
| Education (years) | ||||||||
| ≤13 | 843 | 48.0 | 32 | 42.1 | 470 | 40.7 | 373 | 62.1 |
| 14–17 | 622 | 35.4 | 34 | 44.7 | 430 | 37.2 | 192 | 32.0 |
| ≥18 | 290 | 16.5 | 10 | 13.2 | 255 | 22.1 | 35 | 5.8 |
| Missing | 2 | 0.1 | 1 | 0.1 | 1 | 0.2 | ||
| Income (DM) | ||||||||
| <2567.60 | 579 | 33.0 | 19 | 25.0 | 313 | 27.1 | 266 | 44.3 |
| 2567.60– <3666.70 | 568 | 32.3 | 32 | 42.1 | 366 | 31.7 | 202 | 33.6 |
| ≥3666.70 | 553 | 31.5 | 23 | 30.3 | 440 | 38.1 | 113 | 18.8 |
| Missing | 57 | 3.2 | 2 | 2.6 | 37 | 3.2 | 20 | 3.3 |
Associations between shift work and prostate cancer incidence are reported in table 2. In the fully adjusted model including SES indicators, we observed increased HR among shift workers (HR 2.29, 95% CI 1.43–3.67) and night workers (HR 2.27, 95% CI 1.42–3.64). Being employed for ≥20 years in shift or night work was associated with three- to fourfold increased risks (HR 3.08 95% CI 1.67–5.69 and HR 3.76, 95% CI 2.04–6.93, respectively). The analysis of men employed in shift work never entailing night work showed slightly smaller HR. Because most exposed men (78%) had exposure to both shift and night work, the potential to assess their independent effects was limited and based on only six men with prostate cancer (table 2). Including body mass index, physical activity, and alcohol consumption as confounders changed the overall results only slightly (not shown).
Table 2
Associations between shift and night work in the Heinz Nixdorf Recall Cohort [HR=hazard ratio; 95% CI=95% confidence interval]
| Factor | Person-years | Prostate cancer (N) | HRa | 95% CI | P for trend | HRb | 95% CI | P for trend | HRc | 95% CI | P for trend |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 0–<1 year of shift work | 12 168 | 38 | 1.0 | Reference | 1.0 | Reference | 1.0 | Reference | 0.0001 | ||
| Ever ≥1 years of shift work | 6154 | 38 | 2.0 | 1.28–3.14 | 2.15 | 1.36–3.38 | 2.29 | 1.43–3.67 | |||
| 1–<10 years of shift work | 2500 | 13 | 1.72 | 0.92–3.23 | 0.0012 | 1.82 | 0.97–3.42 | 0.0004 | 1.87 | 0.99–3.55 | |
| 10–<20 years of shift work | 1312 | 8 | 2.03 | 0.95–4.35 | 2.05 | 0.96–4.41 | 2.18 | 1.01–4.72 | |||
| ≥20 years of shift work | 2245 | 17 | 2.38 | 1.34–4.21 | 2.66 | 1.49–4.77 | 3.08 | 1.67–5.69 | |||
| 0–<1 year of shift work | 12 168 | 38 | 1.0 | Reference | 1.0 | Reference | 1.0 | Reference | |||
| Ever ≥1 year of shift work without night work during work life | 1343 | 6 | 1.54 | 0.65–3.64 | 1.57 | 0.66–3.72 | 1.91 | 0.79–4.64 | |||
| 0–<1 year of night work | 13 511 | 44 | 1.0 | Reference | 1.0 | Reference | 1.0 | Reference | <0.0001 | ||
| Ever ≥1 year of night work | 4811 | 32 | 2.01 | 1.28–3.18 | 2.18 | 1.37–3.45 | 2.27 | 1.42–3.64 | |||
| 1–<10 years of night work | 2089 | 11 | 1.60 | 0.83–3.11 | 0.0003 | 1.70 | 0.88–3.30 | <0.0001 | 1.72 | 0.88–3.35 | |
| 10–<20 years of night work | 1031 | 5 | 1.48 | 0.59–3.74 | 1.59 | 0.63–4.01 | 1.68 | 0.66–4.26 | |||
| ≥20 years of night work | 1606 | 16 | 2.99 | 1.68–5.29 | 3.32 | 1.86–5.95 | 3.76 | 2.04–6.93 |
The most frequent jobs associated with shift-work history were in mining [N=45 (7.5%), prostate cancer cases N=5], engine building/production engineering [N=41 (6.8%), prostate cancer cases N=3], and metal working (eg, metal-cutting, metal-forming, foundries) [N=38 (6.3%), prostate cancer cases N=5], which were associated with HR of 2.34 (95% CI 0.92–5.96), HR 1.47 (95% CI 0.46–4.71), and HR 3.68 (95% CI 1.46–9.27), respectively (fully adjusted model). Shift work in industries was associated with an HR 2.45 (95% CI 1.46–4.09) based on 22 prostate cancer cases. Public service jobs involving shift work showed HR 2.26 (95% CI 0.90–5.63) based on five men with prostate cancer who were employed in these jobs (supplemental table S4, www.sjweh.fi/index.php?page=data-repository).
Stratification by preferred midpoint of sleep revealed a trend with shift- and night-work duration particularly for the early sleep preference. We observed smaller, but still increased HR for intermediate and late sleep preferences (table 3).
Table 3
Associations between shift work and prostate cancer according to preferred midpoint of sleep. [HR=hazard ratio; 95% CI=95% confidence interval]
| Factor | Person- years | Prostate cancer (N) | HR a | 95% CI | P for trend | HR b | 95% CI | P for trend | HR c | 95% CI | P for trend |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Preferred midpoint of sleep d | |||||||||||
| Early | 2475 | 13 | 1.18 | 1.20 | 0.64–2.23 | 1.17 | 0.63–2.18 | ||||
| Intermediate | 9829 | 42 | 1.0 | Reference | 1.0 | Reference | 1.0 | Reference | |||
| Late | 2674 | 8 | 0.65 | 0.31–1.39 | 0.70 | 0.34–1.50 | 0.69 | 0.32–1.48 | |||
| Early | |||||||||||
| 0–<1 year of shift work | 1616 | 4 | 1.0 | Reference | 1.0 | Reference | 1.0 | Reference | |||
| Ever, ≥1 year of shift work | 859 | 9 | 4.56 | 1.40–14.89 | 4.14 | 1.21–14.21 | 5.47 | 1.45–20.71 | |||
| 1–<10 years of shift work | 383 | 4 | 4.52 | 1.12–18.34 | 0.0155 | 3.14 | 0.68–14.60 | 0.0150 | 4.09 | 0.80–20.97 | |
| ≥10 years of shift work | 476 | 5 | 4.59 | 1.23–17.14 | 5.05 | 1.33–19.23 | 6.94 | 1.55–31.13 | |||
| 0–<1 year of night work | 1865 | 6 | 1.0 | Reference | 1.0 | Reference | 1.0 | Reference | 0.0031 | ||
| Ever, ≥1 year of night work | 610 | 7 | 3.72 | 1.25–11.10 | 4.68 | 1.44–15.21 | 6.43 | 1.81–22.80 | |||
| 1–<10 years of night work | 239 | 2 | 2.60 | 0.52–13.0 | 0.0117 | 2.93 | 0.54–15.77 | 0.0047 | 4.48 | 0.74–27.11 | |
| ≥10 years of night work | 372 | 5 | 4.48 | 1.37–14.73 | 5.98 | 1.71–21.0 | 7.72 | 1.92–30.97 | |||
| Intermediate | |||||||||||
| 0–<1 year of shift work | 6752 | 20 | 1.0 | Reference | 1.0 | Reference | 1.0 | Reference | 0.0079 | ||
| Ever, ≥1 year of shift work | 3078 | 22 | 2.23 | 1.22–4.09 | 2.36 | 1.28–4.35 | 2.37 | 1.26–4.45 | |||
| 1–<10 years of shift work | 108 | 8 | 2.25 | 0.99–5.13 | 0.0112 | 2.31 | 1.01–5.26 | 0.0071 | 2.26 | 0.98–5.19 | |
| ≥10 years of shift work | 1914 | 14 | 2.29 | 1.16–4.55 | 2.45 | 1.22–4.09 | 2.52 | 1.23–5.19 | |||
| 0–<1 year of night work | 7467 | 24 | 1.0 | Reference | 1.0 | Reference | 1.0 | Reference | 0.0151 | ||
| Ever ≥1 year of night work | 2362 | 18 | 2.12 | 1.15–3.91 | 2.26 | 1.21–4.20 | 2.30 | 1.22–4.35 | |||
| 1–<10 years of night work | 974 | 8 | 2.26 | 1.01–5.05 | 0.0238 | 2.33 | 1.04–5.22 | 0.0150 | 2.38 | 1.06–5.39 | |
| ≥10 years of night work | 1344 | 10 | 2.09 | 1.00–4.38 | 2.26 | 1.07–4.79 | 2.31 | 1.07–4.99 | |||
| Late | |||||||||||
| 0–<1 year of shift work | 1794 | 5 | 1.0 | Reference | 1.0 | Reference | 1.0 | Reference | |||
| Ever, ≥1 year of shift work | 880 | 3 | 1.29 | 0.30–5.44 | 1.23 | 0.29–5.20 | 1.20 | 0.27–5.29 | |||
| 0–<1 year of night work | 1938 | 5 | 1.0 | Reference | 1.0 | Reference | 1.0 | Reference | |||
| Ever, ≥1 year of night work | 736 | 3 | 1.63 | 0.39–6.87 | 1.60 | 0.38–6.71 | 1.42 | 0.33–6.20 | |||
No differences in prostate cancer risk due to shift or night work were seen in the analysis stratified by vitamin D level (table 4).
Table 4
Associations between shift work and prostate cancer according to vitamin D status.
| Factor | Person- years | Prostate cancer (N) | HR a | 95% CI | HR b | 95% CI | HR c | 95% CI |
|---|---|---|---|---|---|---|---|---|
| “Low” vitamin D | ||||||||
| 0–<1 year of shift work | 5161 | 13 | 1.0 | Reference | 1.0 | Reference | 1.0 | Reference |
| Ever, ≥1 year of shift work | 2453 | 13 | 2.12 | 0.98–4.58 | 2.21 | 1.01–4.87 | 2.42 | 1.06–5.51 |
| 0–<1 year of night work | 5663 | 15 | 1.0 | Reference | 1.0 | Reference | 1.0 | Reference |
| Ever ≥1 year of night work | 1951 | 11 | 2.03 | 0.93–4.43 | 2.08 | 0.94–4.63 | 2.22 | 0.98–5.04 |
| “High” vitamin D | ||||||||
| 0–<1 year of shift work | 5072 | 16 | 1.0 | Reference | 1.0 | Reference | 1.0 | Reference |
| Ever, ≥1 year of shift work | 2768 | 17 | 2.04 | 1.03–4.05 | 2.19 | 1.10–4.37 | 2.41 | 1.18–4.92 |
| 0–<1 year of night work | 5726 | 18 | 1.0 | Reference | 1.0 | Reference | 1.0 | Reference |
| Ever ≥1 year of night work | 2114 | 15 | 2.31 | 1.16–4.59 | 2.45 | 1.23–4.90 | 2.63 | 1.29–5.35 |
Analyzing the associations with time to event as timescale did not change the results substantially (supplemental tables S1 and S2). When we restricted the analysis to men participating in the follow-up survey, we observed HR that were similar as compared to the full analysis cohort (results not shown).
Discussion
Using data from the prospective HNR, we identified increased risks for prostate cancer among men who were employed in shift or night work. Prostate cancer risks were particularly pronounced for early sleep preference.
Strengths of this analysis include that we adapted questions from a shift-work module, which was created according to recommendations by an IARC working group and included detailed information to differentiate between shift and night work (16). In contrast to many earlier publications, we were able to consider a large number of potential confounders in our analyses (such as physical activity, family history of prostate cancer, alcohol consumption, and smoking behavior). Furthermore, we were able to test the influence of preferred midpoint of sleep and low vitamin D levels on prostate cancer risk, which are discussed as possible effect modifying factors (16, 20).
Limitations include that the number of incident cases was still small, stratified analyses by duration of shift or night work could not be performed in all subgroups, and the numbers of subjects in subgroups were small and results therefore tended to show wide confidence intervals.
Further, shift work was assessed retrospectively, which may introduce the possibility of reverse causation bias. Reverse causation may occur if prodromal symptoms of prostate cancer affect sleep patterns and subsequently influence the choice of shift worked. We consider the choice of shift work type due to sleep disturbances rather unlikely because the lifelong shift-work history was assessed before baseline, whereas incident cancer was monitored prospectively from the baseline interview. However, we cannot rule out completely that urinary tract symptoms such as nocturia may have modified shift-work exposure as the induction time of prostate cancer is rather lengthy.
It is noteworthy that we observed a much higher prostate cancer risk among men who were not included in the analysis cohort, mainly due to loss to follow-up. Non-participants tended to be older, had lower educational levels, and lower income. Although age may explain part of these differences, a spurious protective association with low SES indicators would rather overestimate the association between shift work and prostate cancer risk. We therefore analyzed a separate model to assess the influence of age, income, education and smoking on prostate cancer risk in the entire HNR cohort (N=2356 men), which showed a decreased HR for low income (HR 0.76, 95% CI 0.48–1.18) and smoking (former smokers: HR 0.60, 95% CI 0.40–0.90; current smokers: HR 0.60, 95% CI 0.35–1.02), but a small increase for low educational level (HR 1.15, 95% CI 0.77–1.74, results not shown). Excluding income and education from the model (equivalent to the absence of confounding by SES indicators) still yielded estimates which were elevated two- to threefold (see table 2). Although we cannot predict a further attenuating effect on risk estimates if low SES indicators were true risk factors for prostate cancer, it would require very strong confounding to reduce these associations to a null effect.
Furthermore, we were not able to include more detailed information on shift work as recommended by IARC, such as time schedules of different shift systems, number of days off after shifts, or direction of shift rotation. These questions were either insufficiently answered in our questionnaire or hard to classify retrospectively due to the complexity of reported shift systems.
It has been pointed out that even early day shifts may be associated with significant circadian disruption in some workers, posing them at increased cancer risk (21). This possibility was implied by our finding that ever shift work, defined as working outside regular hours between 07:00–18:00 hours, showed similar risk estimates to night work taking place at work hours involving substantial circadian disruption.
To assess circadian disruption among shift workers by current diurnal preference is difficult because preferences may change with age. To estimate individual diurnal preference, we solicited men’s preferred midpoint of sleep during leisure days at the 2011 follow-up, which is not an ideal approach (22). Because the subjects’ age in the HNR was >55 years in the follow-up survey, we only determined the current age-specific sleep preference and did not assess the subjects’ past preferred midpoint of sleep. However, the majority of men in the HNR were already retired, limiting entrainment of the circadian clock by occupational and environmental influences (22).
Early reports on the association between shift work and the risk of prostate cancer found heterogeneous results (23–27), but many of these early studies were hampered by methodological concerns, such as misclassification of shift-work exposure and insufficient confounder control (27, 28). However, more recently several studies have been published which support our findings. A Canadian case–control study reported increased odds ratios (OR) for prostate cancer among men performing night work (OR 2.77, 95% CI 1.96–3.92). Estimates were increased across all categories of cumulative duration of night work, although a clear time-dependent trend was not seen (12). In a Spanish case–control study increased prostate cancer risk estimates after 28 years of night work (OR 1.37, 95% CI 1.05–1.81) were reported, but the authors did not observe clear risk differences between workers performing permanent or rotating night shifts (11). Further, the Age, Gene, Environment Susceptibility (AGES) cohort from Iceland showed a two-fold increased prostate cancer risk for men who were suffering from sleep disruption (10). The recent Older Finnish Twin cohort observed an increased risk of prostate cancer among rotating shift workers who were classified as evening chronotypes. In contrast to our results, morning types rather showed a decrease in prostate cancer risk (13). However, the Finnish cohort used self-assessed chronotype, which is not equivalent to the assessment of preferred sleep midpoint and limits direct comparison of these results.
The US Cancer Prevention cohort II observed moderately increased relative risks (RR) for fatal prostate cancer with fixed afternoon/evening shift systems (RR 1.27, 95% CI 0.97–1.65) in the most recent follow-up (1991–2010), whereas rotating shift systems showed RR 1.11, 95% CI 0.97–1.26. The study identified increased risks with insomnia or short sleep duration only in the first, but not the second follow-up, though (7). However, this study concentrated on fatal prostate cancer and therefore does not allow the consideration of less aggressive cancer forms and their association with proxies of circadian disruption.
A large industrial cohort from the BASF company in Germany that did not identify a difference in prostate cancer risk between rotating shift and day time workers is in contrast to our findings. Notably, standardized incidence ratios (SIR) in this cohort were increased in both, rotating shift and day workers (SIR 1.44 and 1.51, respectively) (8). An advantage of this study certainly is that shift schedules at BASF are uniformly fast forward-rotating, which is not the general rule in all industries. In contrast to the BASF cohort, men in the HNR were employed in many different jobs. The most frequent jobs were mining, metal work, and production engineering, which are associated with a variety of shift systems.
Several mechanisms have been discussed regarding the association between shift work and prostate cancer. The “LAN” hypothesis postulates an effect on hormone levels due to circadian disruption and subsequently an influence on sex-hormone levels and hormone-dependent cancers. Much of the research with respect to circadian disruption so far focused on female breast cancer, whereas relatively little research investigated possible mechanisms in male cancers. In support of the LAN-hypothesis, a case–control study nested in the Icelandic AGES cohort found that low first morning void urinary 6-sulfatoxymelatonin levels were associated with sleep problems as well as the risk for prostate cancer (9). Similarly, a cross-sectional survey from the US National Health and Nutrition Survey (NHANES) observed an association between increased prostate-specific antigen (PSA) levels and night or rotating shift schedule, which may indicate an increased risk for future and aggressive prostate cancer (30).
Low vitamin D levels are discussed as possible effect modifying or mediating factor on cancer risk (16, 20). A recent survey from Italy identified lower vitamin D levels among 96 shift workers as compared to 100 workers engaging in regular daily shifts (31). We studied the influence of vitamin D-deficiency on the association between shift/night work and prostate cancer but found that low vitamin D-levels at baseline had no effect modifying effect.
In our data, we observed confounding particularly for low educational levels and low income which both exerted protective effects, thereby increasing the association between shift work and prostate cancer in the fully adjusted model. This rather surprising finding may be due to the fact that subjects of low SES were less likely to participate in the HNR follow-up (see above). Alternatively, we also may have over-ascertained incident prostate cancer among men with high SES (32). In line with this hypothesis, testing for PSA has been demonstrated to be more frequently performed among more affluent men (33, 34), which is important in countries like Germany where PSA testing is generally not covered by the statutory health insurance. Unfortunately, we were not able to investigate further whether lower grade, clinically insignificant cancer was more prevalent among the group of men of higher SES because prostate tumor stage was not solicited in the HNR.
In summary, we identified increased risks for prostate cancer among men engaged in shift or night work. We observed the strongest risks for men with early preferred midpoint of sleep. Smoking behavior and proxies of SES strongly influenced the association between shift/night work and prostate cancer, whereas other lifestyle factors were no important confounders.