Today, women constitute almost half of the workforce in many European countries, and most women work during their reproductive years (1). Studies examining the relationship between employment during pregnancy and reproductive health have yielded contradictory results (2–8). In most European countries, employment during pregnancy selects a population with specific demographic and social characteristics and better access to prenatal healthcare (3). At the same time, work during pregnancy may entail strenuous or hazardous work conditions. A number of chemical and physical agents present in the workplace or related to work organization have been suspected to interfere with the normal reproductive process (9). For some chemicals, such as anesthetic gases, toxic metals, and specific solvents, the evidence is quite compelling (10) whereas for most agents and factors, there is large uncertainty and, if any association exists, it seems of modest magnitude (11). Occupational sectors, for instance the health sector or cleaning work, have the characteristics of employing a large number of women and cumulating several potential hazards according to distinct exposure patterns.
Most of the evidence comes from occupational cohort studies using retrospective data, case–control studies, and registry-based studies (12, 13). The population-based birth cohort design has rarely been used in this field (2, 14–16), mainly because of lack of power when studying rare exposures or occupations and outcomes such as congenital malformations. However, this study design has important strengths such as prospective data collection during pregnancy and potential for controlling relevant confounders. In the present study, we used data from 13 European birth cohorts including >200 000 mother-child pairs to examine whether maternal employment status during pregnancy was associated with birth weight, small for gestational age (SGA), term low birth weight (LBW), length of gestation, and preterm delivery. Among employed pregnant women, we examined these pregnancy outcomes among ten major occupational sectors where exposures potentially hazardous to reproduction are considered to be present. Heterogeneity in effects of all the above-mentioned associations across cohorts was explored. This study is of exploratory nature and the first step towards carrying out further analyses on specific occupational exposures and their potential health effects among children.
Methods
Study populations and data collection
As part of the European-Union-funded Environmental Health Risks in European Birth Cohorts (ENRIECO) project, an inventory was made of environmental exposure data, including maternal occupation, and health data in existing European birth cohort studies (17). Among the 37 cohorts included in the inventory, 15 matched our inclusion criteria of (i) having information on the maternal occupations in the period starting one month before pregnancy until birth and (ii) having coded this information into occupational codes. All these cohorts were invited to participate in this study and 13 cohorts accepted. Of the 2 eligible cohorts that did not participate, 1 declined participation due to a lack of resources and 1 did not reply to the invitation.
A data transfer agreement document was signed by each cohort and datasets were transferred to the Centre for Research in Environmental Epidemiology with personal identifiers removed using a Secure File Transfer Protocol. Informed consent was obtained from all study participants as part of the original studies and in accordance with each study’s institutional review board. The population sample was restricted to live-born singleton infants, with known values for birth weight or length of gestation and available occupational history (N=222 317) (supplemental material table A, www.sjweh.fi/data_repository.php). A live birth was defined as an infant showing signs of life at a length of gestation of ≥22 completed weeks or weighting ≥500 g.
Occupational history
Information whether the mother worked during the period starting one month before conception until birth was obtained by self-reports or from questionnaires carried out by trained interviewers at different time points during pregnancy in each participating cohort. In five cohorts, occupational history covered the whole pregnancy period, including one month before pregnancy, whereas for others this information covered one or two trimesters or birth (table 1). Pregnant women classified as non-employed included those that reported not being employed during the period starting one month before conception until birth but also housewives and students when specified. Self-employed women, for example those working in agriculture, were identified and included as employed.
Table 1
Description of the 13 European birth cohorts: occupational history information and coding. [See page 11 for definition of abbreviations.]
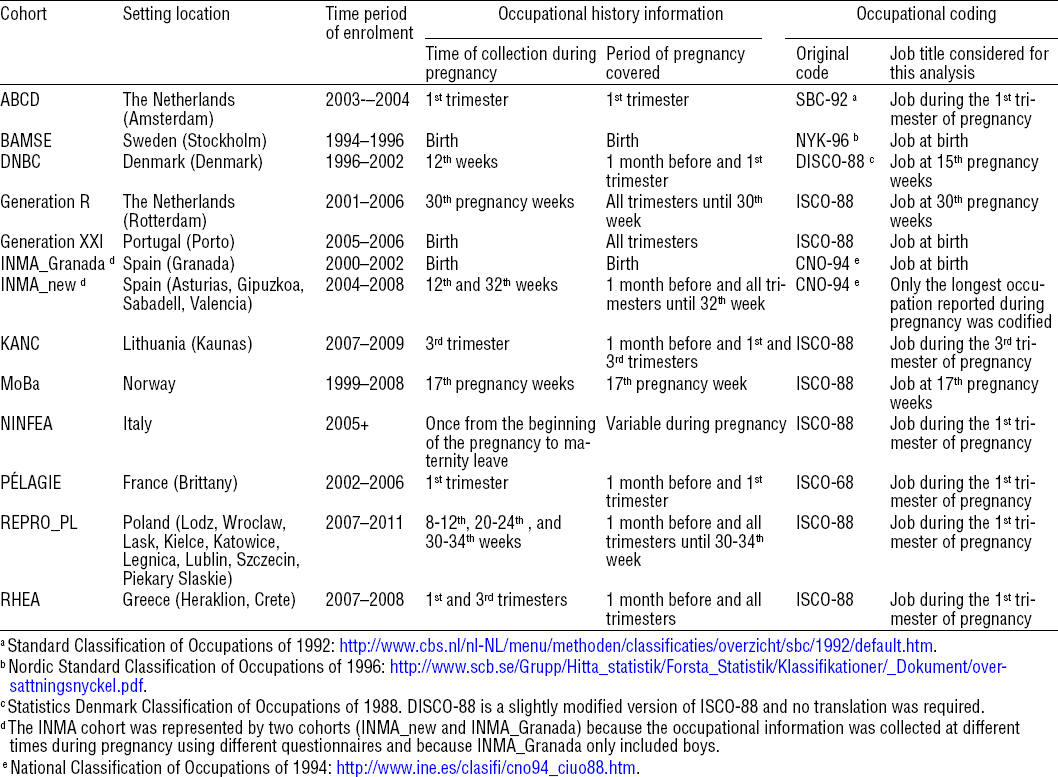
Maternal occupations were coded according to the definition of the International Standard Classification of Occupations of 1988 (ISCO-88) (18). In cohorts where another classification of occupations was used, we translated the codes using the conversion matrices available online into ISCO-88 (table 1). In cohorts where occupational history was collected more than once during pregnancy, only the longest or the occupation reported at the first interview was used (table 1). In cohorts where the mother reported being employed in more than one job at the same time, the first job reported was used in the analysis. Some wrong ISCO-88 codes were replaced by missing codes in job title information.
First, we studied the impact of employment status during pregnancy on birth outcomes comparing employed versus non-employed women. Secondly, considering that an employee can be exposed to a wide range of environmental exposures at the workplace, we defined groups of occupations representing the main sectors of employment among women where potential reproductive hazards are considered to be present (10, 11, 19). Occupational exposures present in these sectors include a variety of chemical, biological and physical factors, together with factors related to work organization. We thus defined ten broad occupational sectors, based on job titles, each sector representing a distinct occupational exposure pattern to chemical, physical and/or biological agents (supplemental material table B, www.sjweh.fi/data_repository.php) including (i) health: infectious agents, pharmaceutical products, ionizing radiation; (ii) daycare: cleaning agents, mild infectious agents; (iii) industry, commerce and home cleaning: cleaning agents; (iv) agriculture and gardening: pesticides; (v) electricity/electronic/optic workers: solvents, paints, glues, inks, welding, brazing; (vi) laboratory work and chemical industry: laboratory and chemical products; (vii) food industry: food additives; (viii) printing and painting: solvents, paints, glues, inks; (ix) hairdressers: cosmetics; and (x) construction workers: solvents, paints. This classification was based on 4-digit ISCO-88 codes except in the Generation XXI cohort where only 3-digit codes were available. A detailed list of the ISCO-88 codes included in each occupational sector can be found in supplemental material table B. Some occupations were classified in more than one occupational sector (eg, “painters and related workers” (ISCO-88: 7141) were classified in the printing and painting and in the construction workers sectors). Within these occupational sectors we further considered certain subsectors or job titles of interest: (i) nurses; (ii) personal care and related workers; (iii) housekeeping and restaurant services; and (iv) domestic and related helpers (supplemental material table B). We selected these subsectors because they accounted for a large proportion of the participants’ occupation in the sector and because previous reports focused on them (20, 21).
Birth weight and length of gestation
Birth weight (in grams) and length of gestation at delivery (in days) were obtained from clinical records (supplemental material tables C1 and C2, www.sjweh.fi/data_repository.php). The last menstrual period (LMP)-based length of gestation estimation was taken if it was consistent by ≤7 days with the ultrasound (US)-based estimation. When both estimations differed by >7 days, or the LMP measurement was unavailable, the US-based estimation was taken. If both measurements (LMP and US) were unavailable, the length of gestation based on maternal report measurement was taken. For the construction of the SGA variable, we first performed a conservative clean-up excluding the most extreme combinations of birth weight and length of gestation (N=37) (22). A SGA binary variable was then defined for each cohort as the 10% with the lowest birth weight stratified by length of gestation, sex of the child, maternal age, and parity. Preterm delivery was defined as birth before 37 weeks of gestation. Term LBW was defined as low birth weights (<2500 g) at term (≥37 weeks) compared to normal birth weights (≥2500 g) at term.
Covariates
Information on tobacco smoking, country of birth, health, education, and other covariates was obtained by self-reported questionnaires, antenatal healthcare visits, or medical/national birth registries (supplemental material tables C1 and C2). Potential confounding variables were defined as similarly as possible among the participating cohorts (supplemental material table D1 and D2, www.sjweh.fi/data_repository.php). For maternal education (supplemental material table E) and maternal country of origin, cohort-specific categories were standardized to create common variables.
Statistical analysis
To assess the association between being employed during pregnancy and birth weight and length of gestation, we used linear or logistic regression models for continuous and binary birth outcomes, respectively; non-employed women were used as the reference group. The same models were used to assess the association between working during pregnancy in a given occupational sector and birth outcomes; in this comparison, the reference group for each specific occupational sector included all pregnant women working during pregnancy except in the occupational sector being assessed (thus including women working in the other nine sectors). Therefore, the size and characteristics of the reference group differed in the analysis of each occupational sector.
We first developed separate regression models for each cohort. Models included subjects with available information on the exposure, outcome and potential confounders; subjects with missing covariates were excluded from the analysis. Models in each cohort were adjusted for maternal age (continuous in years), parity (0, 1, ≥2), maternal education (low, medium, high), maternal smoking during pregnancy (non-smoking, ≤10 and >10 cigarettes/day), maternal body mass index (BMI) (<18.5, 18.5–24.9, 25–29.9, ≥30 kg/m2), marital status (living with the father, living alone), and sex of the child (boy, girl) (supplemental material table D1 and D2). We also adjusted for maternal country of origin (European, non-European) only in cohorts where this information was available and heterogeneous (ie, European and non-European): ABCD, BAMSE, Generation R, INMA_new, NINFEA, and PELAGIE (supplemental material table D1 and D2). Length of gestation was additionally included in the models assessing term LBW. Length of gestation and the square of length of gestation were included in the models assessing birth weight. Models assessing SGA were not adjusted for length of gestation, maternal age, parity, and sex of the child because the SGA definition was based on these variables.
Meta-analyses were used to estimate the overall summary effects of each association. To test the consistency of effect estimates across studies, we used the Cochran’s Q-test (23). If the Q-test was statistically significant (P<0.05), indicating heterogeneity across populations, we used random effects analyses including cohort as random effect and all other covariates as fixed effects. Since the Q-test has low statistical power in meta-analysis with few number of studies we also used the I2 statistic considering an I2≥25% indicating moderate heterogeneity (24). Therefore, when the Q-test was not statistically significant but the I2≥25% we also used random effects analyses. In case of heterogeneity, we used meta-regression to assess whether differences across cohorts were due to the timing during pregnancy when occupational history was collected (whole pregnancy period; 1st, 2nd and 3rd trimester; birth), the geographical region (Southern cohorts: Generation XXI, INMA_Granada, INMA_new, NINFEA, PELAGIE, and RHEA versus Northern cohorts: ABCD, BAMSE, DNBC, Generation R, KANC, MoBa, and REPRO_PL), or the period of enrolment (before or after 2003).
Various sensitivity analyses were performed to assess the robustness of our results. Firstly, to test the influence of the largest cohorts, we repeated the analyses excluding DNBC or MoBa; we also excluded the non-European women in cohorts where this information was available. Secondly, to test whether a high proportion of elected caesarean procedures in a given cohort might explain a reduction in birth weight or length of gestation, we excluded those women with elective caesareans from the models. Thirdly, to test whether maternal diabetes and hypertension during pregnancy could affect the studied associations, we included these variables in the models. Fourthly, potential effect modification by maternal smoking during pregnancy was explored in stratified analyses. Finally, we repeated the models of occupational sectors under study using another group as a reference, defined as working women considered not to be occupationally exposed to reprotoxic agents and with similar socioeconomic status (ie, managers, computer workers, teachers, administrative). Hence, the size of this “unexposed” reference group was the same in all models (N=64 179). This “unexposed” group is potentially more selective, particularly in terms of maternal age, education, smoking, and BMI but not in terms of pregnancy outcomes (supplemental material tables F and G), than the reference groups for the main analysis and therefore was only used for sensitivity analysis. All statistical analyses were conducted with Stata 12.0 statistical software (Stata Corp, LP College Station, Texas).
Results
A total of 222 317 mothers had known values for birth weight or length of gestation and their occupational history data was available. Of these, 177 983 (80%) declared being employed during pregnancy (tables 2a and 2b and supplemental material table A). Among employed pregnant women, 134 312 (76%) had their occupation coded; in MoBa, only 43% of all employees had maternal occupations already coded at the time of the analysis (tables 2a and 2b). Employed pregnant women without coded job title information were excluded from the analysis on occupational sectors.
Table 2a
Distribution of employed women during pregnancy, employees with ISCO-88 codes, and the 10 occupational sectors and subsectors in the total population and each cohort. [See page 11 for acronym definitions.]
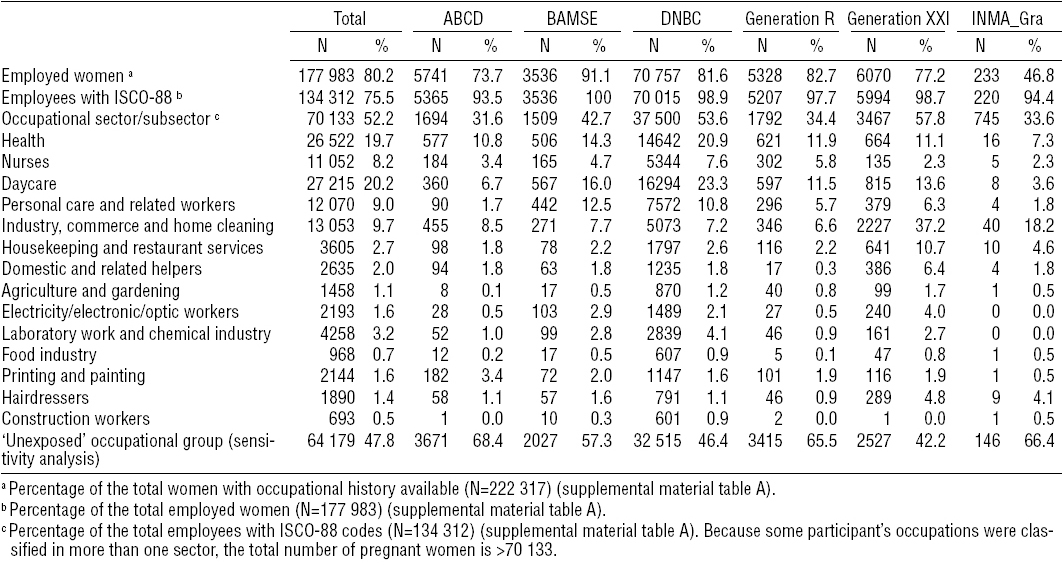
Table 2b
Distribution of employed women during pregnancy, employees with ISCO-88 codes, and the 10 occupational sectors and subsectors in each cohort. [See page 11 for acronym definitions.]
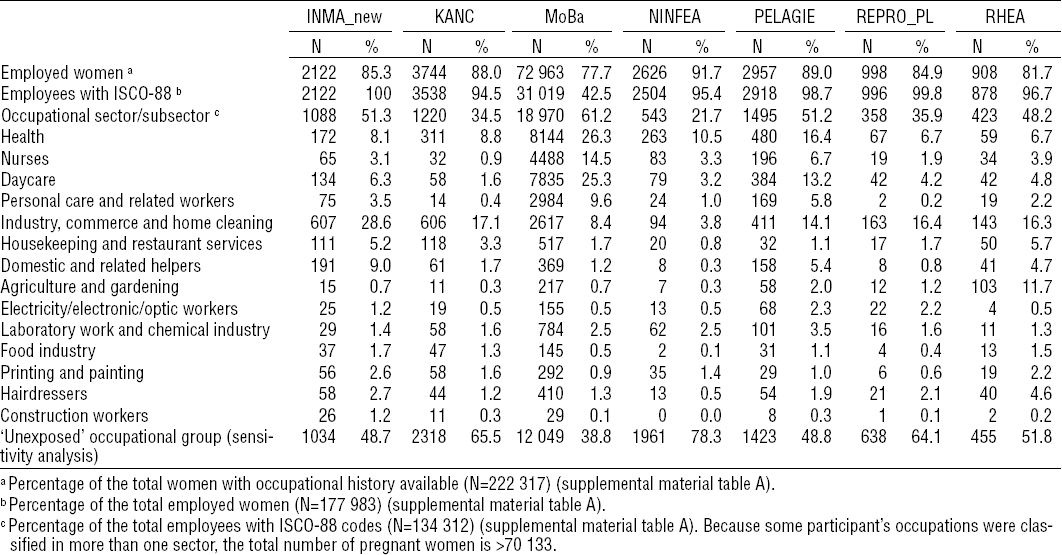
Overall, employed women were older, more likely to be non-smokers, with higher educational levels and lower BMI, and longer length of gestation, compared to women non-employed during pregnancy (supplemental material tables F and G, www.sjweh.fi/data_repository.php). The ten occupational sectors under study accounted for 52% of the employed pregnant women with the highest proportion of women working in the health, daycare, and industry, commerce and home cleaning sectors (tables 2a and 2b). The distribution of these three occupational sectors varied between cohorts: health and daycare were more prevalent in the Northern cohorts whereas industry, commerce and home cleaning were more prevalent in the Southern cohorts (tables 2a and 2b).
Employed women during pregnancy had an average lower child’s birth weight [adjusted beta (βadj) -9, 95% confidence interval (95% CI) 95% CI -14–3], a higher risk of SGA [adjusted odds ratio (ORadj) 1.09, 95% CI 1.02–1.13] and a lower risk of preterm delivery than non-employed women (ORadj 0.86, 95% CI 0.81–0.91) (tables 3a and b, figure 1 and supplemental material figure A, www.sjweh.fi/data_repository.php). Working in most of the ten occupational sectors was not associated with the birth outcomes studied compared to not working in the occupational sector being assessed (tables 3a and 3b). Nurse employment was associated with an average increase in child’s birth weight of 11 g (95% CI 2–21) and a lower risk of the child being SGA (ORadj 0.91, 95% CI 0.84–0.99) (table 3a and supplemental material figure B, www.sjweh.fi/data_repository.php). Personal care and domestic related work was associated with a decrease in child’s birth weight (βadj -21, 95% CI -42–0 and βadj -20 g, 95% CI -40–0, respectively), although the former association was nearly statistically significant (table 3a). Working in the food industry was associated with an average decrease in length of gestation of 1 day (95% CI -1.9– -0.2) and an increase in the risk of preterm delivery (ORadj 1.50, 95% CI 1.12–2.02) (table 3b and supplemental material figure C, www.sjweh.fi/data_repository.php). Working in construction was associated with an average reduction in child’s birth weight of 39 g (95% CI -78–1) and with an increased length of gestation of 1.2 days (95% CI 0.1–2.2) (tables 3a and 3b). No other associations were observed between any other selected occupational sector and birth outcomes (tables 3a and 3b).
Table 3a
Combined adjusted a associations between employed women, occupational sectors/subsectors and birth weight, small for gestational age, and term low birth weight. Effect estimates and cases are presented for the total population. [OR=odds ratio; 95% CI=95% confidence interval. See page 11 for acronym definitions.]
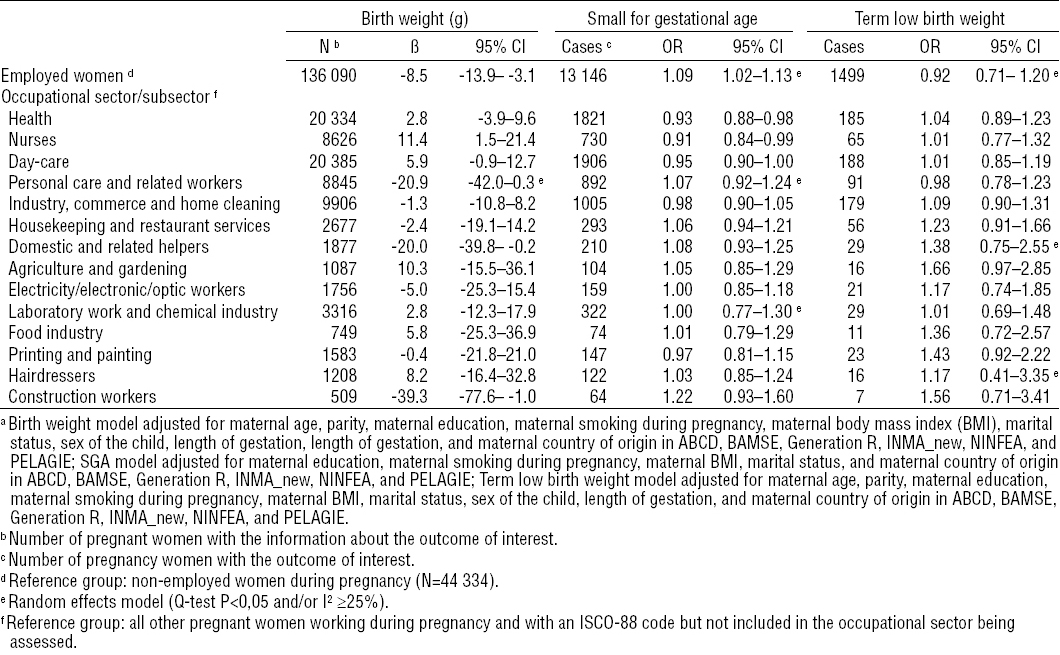
Table 3b
Combined adjusted a associations between employed women, occupational sectors/subsectors and length of gestation and preterm delivery. Effect estimates and cases are presented for the total population. Effect estimates and cases are presented for the total population. [OR=odds ratio; 95% CI=95% confidence interval.]
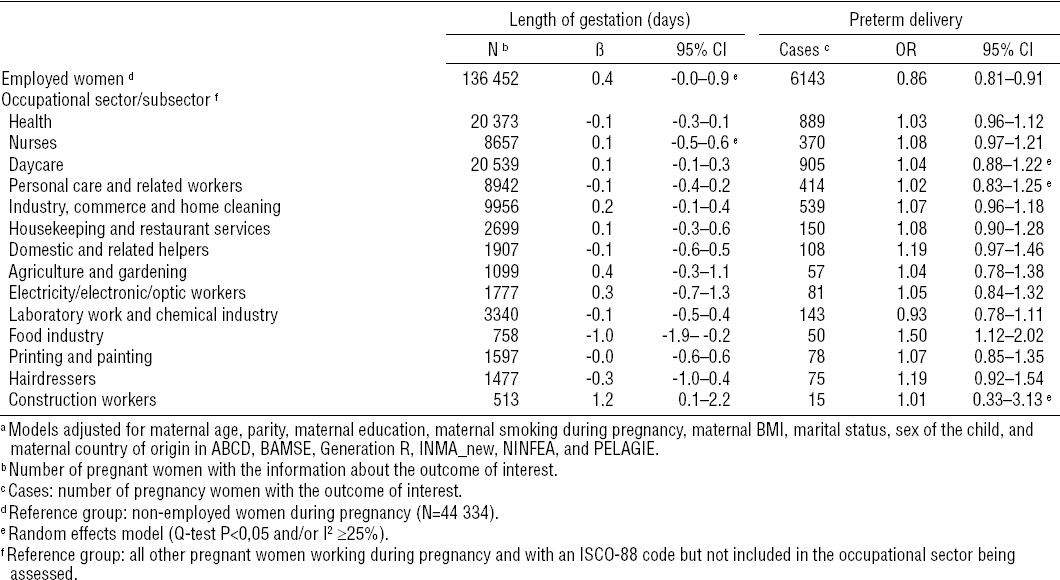
Figure 1
Combined adjusteda associations between employed and non-employed women and preterm delivery. Effect estimates [odds ratio (OR) and 95% confidence interval (95% CI)] and numbers (N) are presented across cohorts and for the total population (Overall) and represent the number of employed women with preterm deliveries. a Adjusted for maternal age, parity, maternal education, maternal smoking during pregnancy, maternal BMI, marital status, sex of the child, and maternal country of origin in ABCD, BAMSE, Generation R, INMA_new, NINFEA, and PELAGIE. See page 11 for definitions of abbreviations.
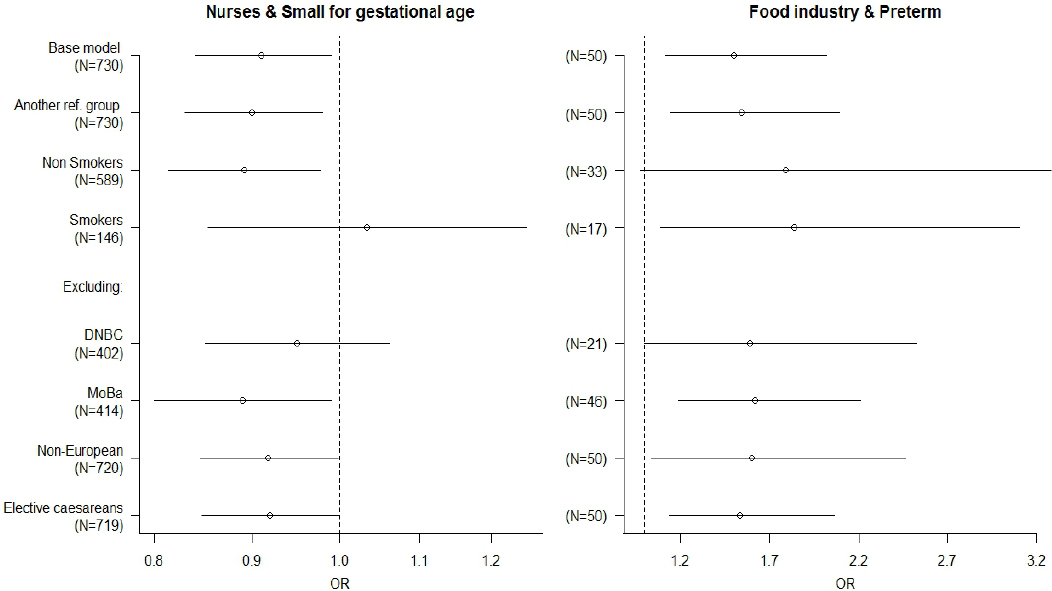
There was little evidence for heterogeneity across cohorts in most of the associations observed. Heterogeneity was only observed in the associations between workers and SGA and between personal care sector and birth weight, but none of the variables tested were explanatory factors of such heterogeneity. After excluding one or the other of the two largest cohorts all the associations observed with birth weight or length of gestation disappeared. The association between construction workers and birth weight was mainly driven by the DNBC cohort, since this cohort represented more than 80% of all construction workers (tables 2a and 2b). The associations observed with SGA or preterm delivery did not change significantly. These associations did not also change after excluding the non-Europeans or elective caesareans (figure 2). Diabetes and hypertension during pregnancy did not change any of the aforementioned associations (data not shown). Moreover, there was no indication of modification by smoking (figure 2). The models of occupational sectors did not change substantially when the “unexposed” reference group was used (figure 2 and supplemental material tables H1 and H2, www.sjweh.fi/data_repository.php).
Figure 2
Sensitivity analyses of the combined adjusted a associations between nurses and small for gestational age and food industry workers and preterm delivery. Effect estimates are presented for the total population. The base model in each association was similar to the one restricted to subjects with information on type of delivery. a Small-for-gestation-age model adjusted for maternal education, maternal smoking during pregnancy, maternal body mass index (BMI), marital status, and maternal country of origin in ABCD, BAMSE, Generation R, INMA_ner, NINFEA, and PELAGIE. Preterm delivery model adjusted for materna age, parity, maternal education, maternal smoking during pregnancy, maternal BMI, marital status, sex of the child, and maternal country of origin in ABCD, BAMSE, Generation R, INMA_new, NINFEA, and PELAGIE. [OR=odds ratio; 95% CI=95% confidence interval. See end of manuscript for definitions of abbreviations.]
Discussion
This large study including more than 200 000 pregnant women of 13 birth cohorts in Europe suggests that employment during pregnancy, as compared to non-employment, is associated with a lower risk of preterm delivery. Working during pregnancy in most occupational sectors is not associated with birth weight or length of gestation compared to not working in the occupational sector being assessed. However, some associations were observed: working as a nurse was associated with favorable birth outcomes whereas food industry employment was associated with less favorable pregnancy outcomes. These findings were consistent across cohorts and across the various birth outcomes studied.
Being healthy is related to access and permanence in the labor market as well as to pregnancy outcome. Results of studies assessing the effects of maternal occupation and pregnancy outcomes are inconsistent with studies showing no indications that prenatal employment status affects birth outcomes (4–8) and others showing an increased risk of adverse birth outcomes among women working >40 hours/week (4, 7, 26). Employed, compared to non-employed, women in our population had a lower risk of preterm delivery, an increased risk of SGA babies and reduced child’s birth weight. However, this reduction in birth weight was only observed in the MoBa cohort and among babies born at term (data not shown). The higher risk of SGA babies was not observed in all cohorts and we could not identify the potential factors of this heterogeneity. In contrast, the association observed with preterm delivery was consistent across cohorts and after performing a series of sensitivity analyses. Studies in Poland and Portugal (3, 6) found an increased risk of preterm delivery among non-employed women, particularly among women seeking employment. Our “non-employed” group was a mixture of women with various statuses, including unemployed, women holding maternity or permanent leave, housewives, and students, and – in most of cohorts – we were not able to separate these groups. It is worth mentioning, that the main group who presented a lower risk of preterm deliveries among employed women was the non-exposed occupational group (ie, managers, computer workers, teachers, administrative). This group had a higher social economic status compared with non-workers and also compared with the majority of the ten occupational groups selected for our analysis. Therefore, the specific association with preterm delivery could indicate that social factors that differ between employed and non-employed are acting and have not been accounted for in the adjustment. This association could be explained by the so-called “healthy worker effect” which means that the observed association might be the result of residual confounding (25).
Among employed women, working as a nurse in our study was associated with favorable birth outcomes. These results contradict most of the previous studies where unfavorable effects among nurses were observed, often in relation with specific exposures (15, 20, 27–29); only one study conducted in Canada showed a lower risk of low birth weight infants among nurses (30). Nurses may engage healthier behaviors during pregnancy than other employees because they have access to preventive recommendations and have better pregnancy follow-up (31). We also found that working in the food industry was related with an increase in preterm delivery and reduced length of gestation. An elevated risk of adverse birth outcomes in these occupations has been described previously (8, 32, 33). Our findings of low birth weight babies and longer length of gestation among women working in construction agree with some epidemiological studies (12, 13, 34), but in our study these associations could only be observed in the largest cohorts, especially in DNBC. We also observed that personal care and domestic related work, more prevalent in the Southern cohorts, was associated with lower child’s birth weight but these associations were not consistent across cohorts and were no longer statistically significant after changing the reference group or after excluding the largest cohort.
Important strengths of this study are its large sample size and the prospective design of the majority of cohorts (in BAMSE, Generation XXI and INMA_Granada data was collected at birth), which makes it less vulnerable to recall bias. We also included a large set of potential confounding factors, usually collected directly from the mother, that have been associated with employment status, occupational exposures, and reproductive outcomes (11). Registry-based studies are usually country-specific and sometimes results cannot be extrapolated to other countries. The present study includes data from 13 countries with a relatively good coverage across Europe and different periods of inclusion, reflecting exposure to job conditions in Europe in the past 1–2 decades. We used occupational sectors, based on job titles, as markers of occupational exposure because information on job tasks and specific hazard exposures in the work setting was not available. Further, most specific job titles did not include sufficient numbers of women. The use of broad occupational sectors may have introduced misclassification as the level/prevalence of exposure to specific hazards may greatly vary between occupations. This may have hampered the detection of associations even in the case of true effects although this was partly avoided when studying occupational subsectors with sufficient numbers and known high levels and prevalence of exposure to specific hazards. Substantial differences in the timing during pregnancy when occupational history was collected were encountered. In cohorts where occupational history only covered one or two trimesters or birth, we assumed that the employment situation remained stable during the whole pregnancy. We considered it unlikely that this assumption affected our findings, since we did not find evidence that this factor predicted heterogeneity across cohorts. Moreover, in cohorts where the mother reported being employed in more than one job at the same time or during pregnancy, the first job reported was used in the analysis. This assumes that the second job reported will be less hazardous for fetal growth which seems reasonable since change in occupation during pregnancy will usually go towards less hazardous exposure. LMP and US measurements are considered the best approaches to estimate length of gestation although they can be subject to recall, misclassification or measurement errors (35). Eleven of our cohorts had information on LMP and/or US for most of the mothers; BAMSE had maternal-based approach as the only measurement and ABCD did not have information on the exact method of estimation.
Baseline participation proportion varied considerably among the different cohorts (from 43–92%). This baseline selection may limit the representativeness to the source populations in some of the studies, especially for the prevalence of the different occupations. However, it is not likely to affect internal validity, ie, the associational estimates between occupational exposures and reproductive outcomes (36), which are the main aim of the current analysis. We need to consider that women not included in our analysis (N=12 897) were more likely to be younger, smokers, and less educated and had less favorable birth outcomes as compared to women included (N=222 317) (data not shown). This has led to an under-representation of pregnant women of the most disadvantaged groups and who are at higher risk of pregnancy complications. We have defined occupational sectors based on potential exposure to chemical, biological, and physical factors but other risk factors related to work organization such as stress, physical load or shift work could also be present and may have had an influence on preterm deliveries in particular. Maternal education was the only indicator of maternal socioeconomic status available and since occupation is strongly related with social position, residual confounding by socio-economic status is likely. The comparison group in our main analysis also included women potentially exposed to environmental hazards at work and this may have led to attenuate associations. Although we have found few associations of modest magnitude, using another reference group in a sensitivity analysis has strengthened our findings. Also, we conducted other sensitivity analyses and evaluated the robustness of results across cohorts and across the various birth outcomes. Multiple comparisons may have given rise to some chance findings in our analyses. Despite the large sample size of the present study, this was still not sufficient to produce stable estimates of associations in occupational strata with few cases of SGA, preterm deliveries, and term LBW.
This collaborative project represents a first step towards carrying out further analyses such as the application of job exposure matrices, which would allow us to assign specific occupational exposures of interest, to a whole range of job titles in population based studies (37, 38).
Concluding remarks
The results of this collaborative effort of European birth cohorts suggest that employment during pregnancy is associated with a reduced risk of preterm birth and that work in certain occupations may be associated with changes in child’s birth weight and length of gestation. This study provides a good picture of the overall impact on the course of pregnancy in important occupational sectors of the women’s workforce in Europe. It provides also a useful platform on which to base further prospective studies of more specific maternal occupational exposures and child health and development after birth.



