Physical activity is generally considered to be good for health, but it has been suggested that physically strenuous activities during pregnancy, including heavy lifting, may increase the risk of fetal death (1–4). Fetal death is defined as the intrauterine death of any conceptus at any time during pregnancy (5).
Until 2009, the Danish Working Environment Authorities stated that pregnant women should not lift loads weighing >12 kg or a total of >1000 kg during a work day (6). These absolute guiding limits were removed due to lack of evidence, and lifting of heavy burdens is now included in the recommendations as part of a broad measure of physical strain together with other biomechanical exposures (7).
Two large pregnancy cohort studies did not find that heavy lifting was associated with miscarriage (8, 9). In contrast, an increased risk of miscarriage was seen with high physical strain around the time of conception in a cohort of first pregnancy planners with daily records of activity level and monitoring of hormonal status (3). The remaining evidence is primarily based on case–control studies and cross-sectional surveys with results pointing in different directions (10–19). Finally, two recent cross-sectional studies from Asia and Africa draw attention to the need for continuous concern for the working environment of pregnant women: a recent cross-sectional study from South Korea supported a potential association between occupational heavy lifting and miscarriage (4), and, in Nigeria, work-related physical exertion was associated with preterm birth while lifting of heavy objects at home was associated with low birth weight (20).
Even though the potential mechanisms between physical activity and fetal death are unknown, such an association is biologically plausible. The physiological changes that occur during physical exertion are associated with an acute reduction in the maternal-fetal blood flow (3, 21–23). In addition to this transitory vascular effect, occupational biomechanical exposures (such as quiet standing, frequent bending, or heavy lifting) may cause protracted reductions in the uterine blood flow, even if the exposures are intermittent (2–4, 20–26).
Due to the likely different aetiologies of early miscarriage, late miscarriage, and stillbirth, the aim of the present study was to examine the association between occupational lifting and the risk of fetal death according to gestational age. We analyzed the daily frequency of occupational lifting, the weight of single loads lifted, and the number of kilos lifted per day using data from a large, population-based, pregnancy cohort.
Methods
We used data from the Danish National Birth Cohort (DNBC), a nationwide population-based cohort of approximately 100 000 pregnancies recruited during 1996–2002. Pregnant women were informed about the study by their general practitioner at the first antenatal care visit, which is usually scheduled during gestational weeks 6–12. In Denmark, antenatal care is part of the public health system and is used by 99% of all pregnant women (S. Rasmussen, National Board of health, personal communication, 2007). Participants were included in the study when their informed consent was registered. Participants were contacted for a computer-assisted telephone interview on background, health, and lifestyle factors, scheduled during gestational weeks 12–16 (ie, pregnancy interview (27). Women, who experienced a fetal death after inclusion in the study, but before the scheduled telephone interview, were asked to answer slightly modified questions in the interview (ie, case interview) (1).
Data from either the pregnancy or the case interview were available for 92 717 out of 100 418 pregnancies initially enrolled in the DNBC. For this study, we restricted the cohort to the 75 602 pregnancies of women who worked ≥15 hours per week by the time of the interview or within the past three months. We excluded ectopic pregnancies and mola hydatidosa events (N=40) and pregnancies with missing or incomplete information on gestational age at recruitment or at pregnancy termination (N=31), occupational lifting (N=1698) or any of the other covariates included in regression models (N=2333). The final study population comprised 71 500 pregnancies of occupationally active women: 69 468 with prospectively collected data and 2032 with retrospectively collected data.
The National Committee on Health Research Ethics has approved the DNBC, and – before initiating the present study – approval by the Danish Data Protection Agency was obtained.
Exposure measurement
The participating women were asked about heavy loads (>20 kg): “In your job, do you daily lift more than 20 kilos at a time?” and, if they answered yes: “How many times a day do you lift more than 20 kilos?” The questions were repeated for medium loads (11–20 kg). We calculated the cumulated number of kilos lifted per day and established the following categories: 0–14, 15–100, 101–200, 201–500, 501–1000, >1000 kg per day. In this calculation, the pre-defined heavy loads were set to 22.5 kg, medium loads to 15 kg, and lifting either of the weight loads or both less than once a day was set to 0–14 kg. The latter is referred to as the non-lifting group. Furthermore, we generated a nine-value variable, which combined the frequency of lifting 11–20 kg and the frequency of lifting >20 kg, using a 3-level frequency measure (0, 1–10, >10 times/day).
During the interview, the trained interviewers assigned a DISCO-88 code (Danish version of the International Standard Classification of Occupations from 1988) to the job title. In case of uncertainty about the reported job title, the interviewer asked additional questions, until a DISCO code was determined. For sub-analyses regarding person-lifting, we defined the following occupations as potentially involving lifting of persons: nurses, nursing assistants, midwives, health visitors, nursery teachers/assistants, hospital porters, and ambulance assistants or paramedics.
Covariates
We decided a priori to adjust for maternal age (<25, 25–<35, 35–<40, ≥40 years), parity (0, ≥1), pre-pregnant body mass index (<18.5, 18.5–<25, 25–<30, 30–<35, ≥35 kg/m2), and the woman’s occupational status (higher grade professional, lower grade professional, skilled worker, unskilled worker, student). Information on occupational status was ascertained from the self-reported job titles, in which the educational level is reflected, combined with information on current job situation. We furthermore adjusted for the following exposures during pregnancy: daily smoking at any time before interview (0, 1–9, ≥10 cigarettes per day), alcohol consumption (0, 0.5–1.5, 2–3.5, ≥4 units per week), leisure-time physical exercise (yes/no), leisure-time daily lifting >20 kg (yes/no), and predominant working posture (primarily standing or walking, primarily sitting, varying).
Outcome measurement
From The Danish Civil Registration System and The Danish Medical Birth Registry, we identified all live births and stillbirths by use of the woman’s civil registration number and the link between mother and child. We obtained information on all other pregnancy outcomes and the gestational age at pregnancy termination based on last menstrual period from the Danish National Patient Register. We divided fetal death into early miscarriage (≤12 gestational weeks, ie, 0–83 days), late miscarriage (13–21 gestational weeks, ie, 84–153 days), and stillbirth (≥22 gestational weeks, ie, ≥154 days).
Statistical methods
We used SAS statistical software V9.2 (SAS Institute, Cary, NC, USA) to conduct Cox regression analysis with number of days since last menstrual period as the underlying time variable. We included early and late miscarriages and stillbirths as events and censored live births, early induced abortions, late induced abortions (fetal or maternal indication), emigration, and maternal death during pregnancy. For most of the included covariates, the proportional hazards assumption was not fulfilled, so early and late miscarriage and stillbirth were analyzed separately. Women, who consented before day 84 were included in early miscarriage analyses, where follow-up started at time of consent and ended at pregnancy termination, emigration, maternal death, or completion of gestational week 12, whichever came first. Women, who were enrolled before day 154 and still pregnant at day 84, were included in late miscarriage analyses, where follow-up started at time of consent or at day 84, whichever came last, and ended at pregnancy termination or completion of gestational week 21, whichever came first. All women, who were still pregnant at day 154 were included in stillbirth analyses. Follow-up started at time of consent or at day 154, whichever came last, and ended at pregnancy termination. Because of few events in some cells, some of the higher lifting groups were collapsed in the analyses of late miscarriage and stillbirth.
First, we calculated the hazard ratios (HR) of fetal death according to (1) the total number of kilos lifted per day, (2) the number of single lifts of 11–20 kg and >20 kg per day, and (3) a combined measure of lifting frequency and number of kilos. We stratified our models by gestational age at the time of consent. Some women participated with more than one pregnancy, so we calculated robust sandwich standard errors to account for the within-person correlation between pregnancies. Second, we included the possible confounders. Third, we repeated the analyses of late miscarriage and stillbirth on the subset of pregnancies with prospectively collected data. In this model, we stratified by time of interview, and follow-up started at time of pregnancy interview. Due to few early spontaneous abortions in the prospective data set, these analyses were carried out only for late spontaneous abortions and stillbirths. We calculated P-values for trend for amount and frequency of occupational lifting. Finally, we compared the likelihood of reporting occupational lifting among women interviewed before and after a fetal death, given by (N, exposedprospective data/N, unexposedprospective data)/(N, exposedretrospective data/N, unexposedretrospective data) among women with a fetal loss during gestational weeks 11–17 (ie, an overall estimate for the period and separate estimates for each week), including a test for difference between weeks.
We repeated main analyses restricted to first-time pregnant women to avoid the influence of a woman’s reproductive history, and to singleton pregnancies because of the correlation with both occupational behavior and the course and outcome of pregnancy. Finally, we analyzed kilos lifted per day according to lifting of persons using an 11-value variable with non-lifters as reference and categories of kilos per day for jobs including and not including the lifting of persons.
Results
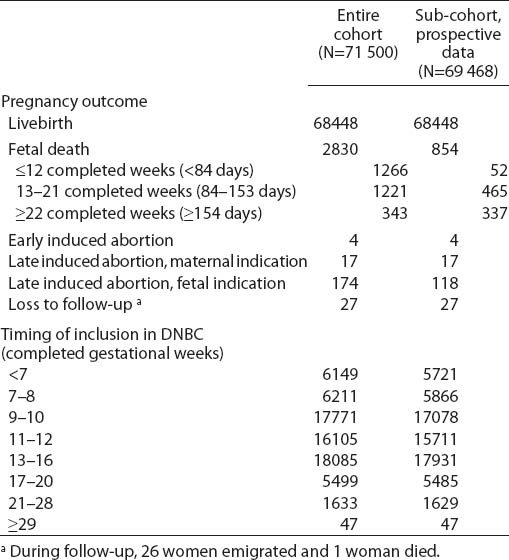
Of the 71 500 pregnancies, 2830 ended with a fetal death. One third of the events occurred after the baseline interview. Hence, out of 1266 early miscarriages, prospectively collected data were available for 52 (4.1%). For late miscarriages and stillbirth, these proportions were 38% and 98%, respectively (table 1).
Table 1
Pregnancies in the study according to pregnancy outcome and time of inclusion in The Danish National Birth Cohort (DNBC) 1996–2002. N=71 500.
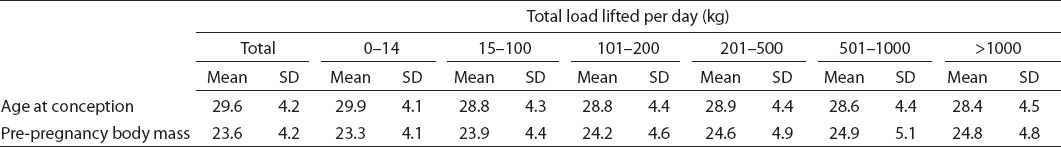
Women with occupational lifting more often held unskilled jobs and had standing or walking as main working posture compared with non-lifters (table 2a). They were more likely to have given birth before, smoke during pregnancy, and report heavy lifting outside work, but less likely to engage in physical exercise. Moreover, women with occupational heavy lifting were slightly younger and had a slightly higher body mass index than non-lifters (tables 2a and 2b).
Table 2a
Maternal characteristics according to occupational heavy lifting during pregnancy. The Danish National Birth Cohort 1996–2002. N=71 500.
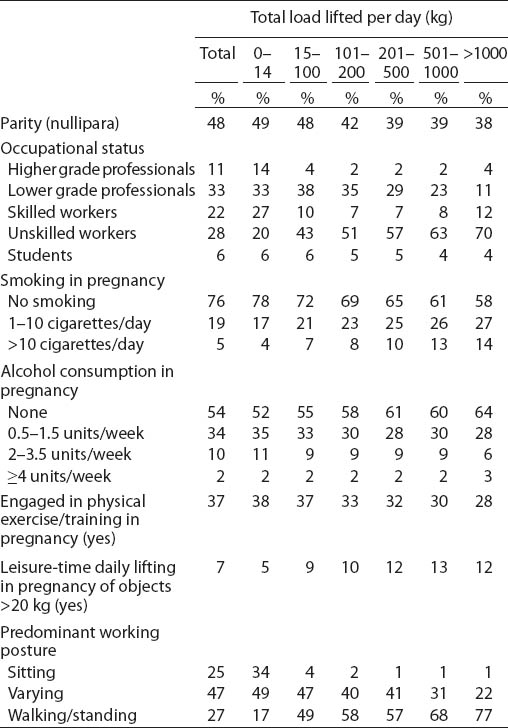
Table 2b
Maternal characteristics according to occupational heavy lifting during pregnancy. The Danish National Birth Cohort 1996–2002. N=71 500. [SD=standard deviation]
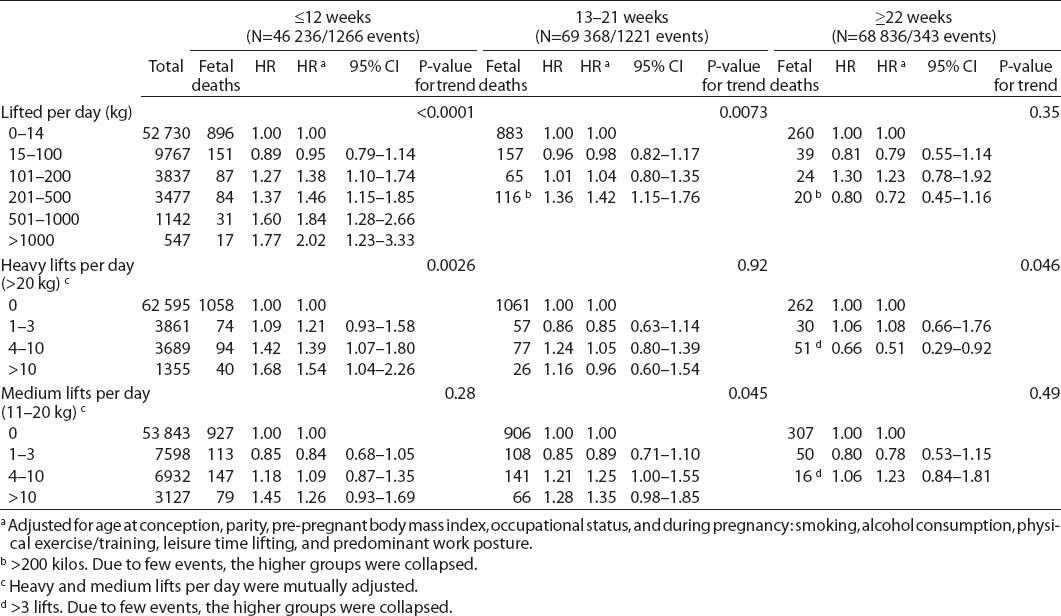
Just over one fourth of the women lifted loads >10 kg on a daily basis at work, 12% lifted loads >20 kg, and 13% lifted >100 kg per day in total (table 3). The risk of early miscarriage increased with increased total burden lifted per day (P<0.0001), eg, women, who lifted >1000 kg per day had a doubled miscarriage risk as compared to non-lifters after adjustment for confounders [HR 2.02, 95% confidence interval (95% CI) 1.23–3.33]. The risk of a late miscarriage was higher among women lifting >200 kg per day than among non-lifters (HR 1.42, 95% CI 1.15–1.76). No obvious associations were observed for stillbirth. In the analyses of daily lifting frequency, we found a positive association between number of heavy lifts and early miscarriage and between number of medium lifts and late miscarriage (table 3). Furthermore, there was some indication of an increased risk of fetal death during all periods of pregnancy for women with >3 medium lifts per day, but with statistical uncertainty.
Table 3
Adjusted hazard ratios (HR) for early miscarriage, late miscarriage, and stillbirth according to occupational heavy lifting during pregnancy. The Danish National Birth Cohort 1996–2002. N=71 500. [95% CI= 95% confidence interval]
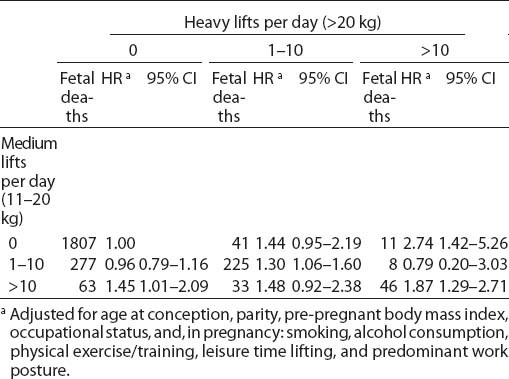
For the combined effect of weight of single loads and lifting frequency on early miscarriage, we found the highest risk among women with >10 heavy lifts combined with no medium lifts (HR 2.74, 95% CI 1.42–5.26) and with >10 medium lifts (HR 1.87, 95% CI 1.29–2.79) (table 4). Thus, there was no clear interaction pattern between the lifting of heavy and medium loads (P=0.36).
Table 4
Adjusted hazard ratios (HR) for early miscarriage according to a combined measure of number of heavy and medium lifts per day. The Danish National Birth Cohort 1996–2002. N=46 236. [95% CI=95% confidence interval]
Restricting data to first-time pregnant women or singleton pregnancies revealed essentially the same results as the main analyses. A restriction to prospectively collected data did not change the results substantially except for reducing the precision of the estimates (see Appendix table A). In concordance with this, our data did not indicate any systematic differences in the reporting of occupational lifting depending on mode of data collection (ie, prospective versus retrospective). Among women with miscarriages, the odds ratio of reporting any daily occupational lifting >10 kg was 1.16 (95% CI 0.89–1.50) for women prospectively interviewed relative to women interviewed after a fetal loss. There was no indication of different effects between the single weeks from 11–17 (P=0.16) (see Appendix table B).
Just over one fourth of the women who reported occupational lifting held jobs that usually involves lifting of persons. This proportion was the same in the prospectively and retrospectively collected data. Overall, there were only slight differences in the association between occupational lifting and fetal death among women in jobs with and without lifting of persons. Yet, compared to no lifting, the HR of lifting more >1000 kg per day was 2.91 (95% CI 1.16–7.29) in jobs with lifting of persons and 1.79 (95% CI 1.00–3.32) in jobs with no lifting of persons (table 5).
Discussion
Women with occupational lifting were more likely to have a first trimester miscarriage than women with no lifting, and the risk increased with total number of kilos lifted per day and with lifting frequency. Late miscarriage risk (13–21 weeks) was increased with total weight load and number of medium heavy lifts per day, but not with number of lifts >20 kg. No association was seen for stillbirth (≥22 weeks). One out of four women in the DNBC reported daily occupational lifting >10 kg.
Contrary to our findings, two cohort studies with early pregnancy recruitment and prospective exposure data did not find occupational lifting associated with miscarriage (8, 9). These older studies (recruitment 1980–1983 and 1990–1991) were carried out in affluent countries with measures of occupational lifting broadly similar to those in the DNBC. Despite apparent similarities, both of these studies used lower categories of weight loads than in the DNBC, which may partly explain the negative findings. In the study by Ahlborg and colleagues, occupational lifting was measured as loads >7 kg, and – in that of Fenster and colleagues – as loads ≥12 kg (8, 9). Similarly, the maximum frequency of lifting was >10 and 15 times per day, respectively. In the DNBC we measured both 11–20 kg and >20 kg as well as the daily frequency. A positive association between high biomechanical load and miscarriage was reported from two other cohort studies characterized by pre-pregnant recruitment and the use of clinical and biological measurements in addition to self-reported exposure information (3, 25). Florack and colleagues found a threefold increased risk of miscarriage with high biomechanical exposures, but the risk was explained by frequent bending rather than heavy lifting (25). Hjollund and colleagues found high physical strain at day 6–9 after ovulation associated with a 2.5–3 fold increased miscarriage risk (3).
The apparently low prevalence of miscarriage (3.5%) and stillbirth (0.5%) in this study reflects the fact that women were enrolled after their first antenatal visit, which means that the cohort did not enroll many pregnancies ending in very early miscarriage. Application of a fetal life-table approach to the data has, however, estimated the cumulative risk of fetal loss from the beginning of week 6 to be 11% (28), which matches national estimates of fetal loss (29). We did not have data to examine whether non-participation differed between women with limited workload and a miscarriage and women with heavy workload and a miscarriage. Although we know of no generic mechanism creating selection bias, it cannot be ruled out with reasonable confidence. Likewise, our data do not allow us to estimate the occupational lifting-related risk of very early fetal loss.
Our data did not demonstrate an association between occupational lifting and stillbirth, which is in line with a comparable Swedish study with recruitment and data collection in early pregnancy (8). Apart from this, we have not identified original papers examining this relation, and, accordingly, stillbirth is not mentioned in review papers on occupational biomechanical exposures and fetal death.
In addition to its size, the present study has several other strengths: (i) linkage to nation-wide registries ensured nearly complete follow-up. The Danish National Medical Birth Registry comprises records on all births in Denmark. (ii) The study recorded a fairly high number of first trimester fetal deaths. (iii) The study included detailed information on a number of risk factors for fetal death allowing for thorough confounder adjustment. (iv) The questions on lifting exposure in the DNBC allowed for separate analyses of the cumulated weight lifted per day and the frequency of lifting.
Information on occupational lifting was self-reported and, thus, vulnerable to bias. It has been suggested that the perception of lifting the same amounts and the same frequency of lifting differs between occupational groups (30). For example, if certain occupational groups tend to overestimate the lifting measures due to a general awareness of potential hazards related to high physical strain, differential misclassification would occur. This has been suggested for women in occupations that include lifting of persons, such as nurses or nursing aids. Even though our data did not reveal any convincing differences in early miscarriage risk between women in jobs with and without lifting of persons, there was an almost three times increased risk with lifting >1000 kg per day from tasks involving lifting of persons. This may reflect a cultural phenomenon in the reporting of lifting, as suggested above, or it may be explained by differences in the mechanical character of the lifting procedures when lifting persons compared to other objects, eg, differences in the leverage of the lifting, lifting techniques, the use of devices in lifting, lifting of dead versus living material, lifting close to the body etc. We believe this to be an important focus for future studies on occupational hazards among pregnant women.
By design, the analyses on early miscarriage were based mainly on retrospectively collected exposure data. Due to the narrow time window for data collection from conception to pregnancy termination, recruitment should ideally take place before conception to avoid recall bias. This is time and finance consuming, and, accordingly, very few pre-pregnancy cohorts exist (3, 25). To investigate the magnitude of recall bias, we compared the reporting of occupational lifting among women interviewed before versus after a fetal death, and we repeated the main analyses on a dataset restricted to prospectively collected data only. This investigation did not indicate systematic differences in lifting information according to mode of data collection, and there were no considerable changes in risk estimates from the prospective data as compared with those from the whole population. The finding is accordance with the observations by Fenster et al (9).
In this study, we focused on heavy lifting as one important occupational exposure and adjusted for leisure-time physical activity. Comparisons between studies are complex though because of a substantial variation in exposure measurement; occupational biomechanical exposures include several specific routines, such as lifting, bending or reaching, and these exposures are further part of the overall physical activity measure, which also counts household tasks and recreational exercise. In general, the scientific literature points toward adverse effects of occupational-related physical activity and healthy effects of recreational physical activity, suggesting that adverse pregnancy outcomes related to occupational exposures may be related to the specific character of the activities rather than the physical activity per se (23). Standing and walking at work as well as leisure-time heavy lifting correlated with occupational heavy lifting in our descriptive analyses, but including these two measures in the regression model did not noticeably affect the association between occupational lifting and fetal death. They were, however, included in the final model due to the a priori selection of adjustment variables.
Occupational lifting was not associated with stillbirth in the current study, and one explanation might be that the women in the DNBC were not, or only minimally, exposed in the last part of pregnancy. If some women reported lifting in the early pregnancy interview, but reduced or ceased lifting later in pregnancy, this would weaken an association with stillbirth. This could be the case as a consequence of Danish legislation, which entitles pregnant women to paid absence if their work involves a risk to the fetus and referral to suitable alternative work procedures is not possible. Such preventive efforts might explain why we found a higher risk of mid-pregnancy fetal death with lifting medium (11–20 kg) than heavy (>20 kg) loads. The positive association between lifting and fetal death in early pregnancy, where the majority of lifting data were collected, together with an assumption of a gradual decline in occupational lifting as pregnancy develops is consistent with a short lag time hypothesis between exposure and fetal death. Since we did not have gestational age-specific lifting data, this study cannot rule out an association between lifting late in pregnancy and stillbirth.
In conclusion, our findings support an exposure–response relation between occupational lifting and miscarriage, particularly in the first trimester. We found no association with stillbirth. The results need cautious interpretation because of incomplete ascertainment of miscarriage. If our findings are corroborated, there may be a need for intensified efforts to reduce occupational lifting early in pregnancy.




